|
|
||||
|
Published by : PROFESSIONAL MEDICAL PUBLICATIONS |
||||
|
ISSN 1681-715X |
||||
|
||||
|
- |
||||
|
ORIGINAL ARTICLE |
||||
|
- |
||||
|
Volume 22 |
January March 2006 |
Number 1 |
||
|
|
||||
|
|
||||
|
|
||||
|
Published by : PROFESSIONAL MEDICAL PUBLICATIONS |
||||
|
ISSN 1681-715X |
||||
|
||||
|
- |
||||
|
ORIGINAL ARTICLE |
||||
|
- |
||||
|
Volume 22 |
January March 2006 |
Number 1 |
||
|
|
||||
|
|
||||
How do the intercept and the gain of the dyspnea -
The heart rate relationship respond to exercise
in cardiac patients?
Shabbir Bokhari1, Asad Ali2, Feisal Subhan3,
ABSTRACT:
Objectives: To study the dyspnea-heart rate relationship in patients with a positive exercise tolerance testing (ETT) and to compare it with normal healthy controls who had a negative ETT.
Design: This was a matched case-control study, in which all patients and their controls underwent an ETT (Bruce protocol) at the Cardiopulmonary Diagnostics Laboratory of a tertiary health care facility.
Methods: Twenty two male patients, who tested positive on an ETT, were taken as cases and their dyspnea profile was measured during the ETT using the Borg scale. Age, sex and body mass index matched healthy subjects who tested negative on the ETT were recruited as controls and underwent similar dyspnea assessment. Regression analysis of the dyspnea/heart rate relationship was performed to calculate the slope and intercept for every patient and these were compared with the values of the respective controls.
Results: The dyspnea/heart rate relationship in patients was found to be linear. The mean intercept (� SEM) was significantly lower in patients when compared with controls; 100.68 � 5.25 beats min-1 in patients compared with 113.14 � 3.51 beats min-1 in controls (p=0.005). However, the slope did not show any significant difference between patients and controls.
Conclusion: Changes in dyspnea, in this group of patients, is translated via modulation of the intercept (threshold) of the dyspnea/heart rate relationship.
KEYWORDS:
Heart rate, Dyspnea, Angina Pectoris, Exercise tolerance test, Intercept, Slope, Ischemic heart disease.Pak J Med Sci January-March 2006 Vol. 22 No. 1 28-32
1. Dr. Shabbir Bokhari
2. Dr. Asad Ali
3. Dr. Feisal Subhan
4. Dr. Nasiruddin Ahmed
5. Dr. HR Ahmed
1-3&5: Department of Biological and Biomedical Sciences,
The Aga Khan University, Stadium Road,
Karachi-74800, Pakistan
4: Department of Medicine, Faculty of Health Sciences,
The Aga Khan University, Stadium Road,
Karachi-74800, Pakistan
Correspondence:
HR Ahmad, MD PhD Bochum, FCPS hc,
Professor of Physiology & Dean Postgraduate Studies,
Ziauddin University.
4/B Shahrah-e-Ghalib, Clifton, Karachi-75600, Pakistan
E-mail: hrahmad@zmu.edu.pk
* Received for Publication: August 27, 2005
Accepted: November 22, 2005
INTRODUCTION
Dyspnea is a distressing symptom of patients with ischemic heart disease1 and those of pulmonary disease.2 Dyspnea is a significant symptom of undiagnosed Coronary Artery Disease (CAD), when exertional angina and EKG evidence of ischemia are absent.3 Studies in healthy subjects have shown that having a sole symptom of effort-related dyspnea increases the likelihood of dying from coronary disease in the subsequent years.4, 5 Patients who complain of dyspnea have a higher prevalence of angina pectoris; this association is independent of age and cigarette smoking and is related to the severity of dyspnea.3 A significant proportion of angina pectoris patients suffer from dyspnea.1 It has also been shown that dyspnea in cardiac patients correlates better with Left Ventricular Ejection Fraction, as compared to chest pain.6
Exercise tolerance testing (ETT) can be used as a clinically relevant tool to assess dyspnea.7 ETT is commonly employed for diagnosis of CAD8 and to assess the pre-operative fitness of patients undergoing major surgery.9 The results of ETT classify the patient as high or low risk for CAD.8 Since the current protocols for ETT are standardized, it provides an excellent opportunity to study dyspnea and derive clinically pertinent inferences.
The linear nature of the dyspnea-ventilation relationship has been well studied.10 There have been several studies in which the dyspnea-ventilation relationship has been analyzed using the intercept and slope of this relationship.11-14 We have recently shown that the dyspnea-heart rate relationship is as linear as that of the dyspnea-ventilation relationship.15 Since measurement of heart rate is simpler than that of pulmonary ventilation in clinical practice and is regularly performed during an ETT,8 it offers an advantage to study the mechanisms of dyspnea in relation to this cardiac parameter.
This study evaluated the following hypotheses:
a) Is the dyspnea/heart rate relationship linear in patients with who have a positive ETT?
b) How do the intercept and slope of the dyspnea-heart rate relationship respond in these patients relative to normal subjects?
METHODOLOGY
In this matched case-control study, twenty-two subjects were recruited from the Aga Khan University (AKU) Hospital Cardiopulmonary Diagnostics facility. These patients tested positive on an Exercise Tolerance Test (ETT), either with chest pain or electrocardiogram (EKG) changes consistent with ischemia or both. These patients were individually sex, age and body mass index (BMI) matched with 22 healthy controls who performed the ETT for functional capacity studies and who tested negative.
The study was approved by AKU Ethical Committee and performed according to the declaration of Helsinki. After giving informed consent, subjects undertook a maximal progressive exercise test following a Bruce Protocol on a treadmill. Dyspnea was estimated each minute during the test using the modified Borg Scale. The wordings on the Borg Scale were translated into Urdu (the national language of Pakistan), for the ease of study participants although an English version was also provided for those who could understand the language. Blood pressure and a 12 lead EKG were also obtained during the ETT. The end point of the exercise was either achievement of the target heart rate (85% of the predicted maximal heart rate), pain in the chest or arms, leg pain, exhaustion, maximal dyspnea (Borg score of 10) or EKG changes consistent with ischemia.Before the start of each ETT, the term �dyspnea� was defined, in Urdu, to the participants as a sense of air-hunger, a feeling that breathing was not sufficient for the needs they thought they had, or a shortness of breath. It was emphasized that they should not confuse dyspnea (breathlessness) with other sensations associated with exercise, such as leg fatigue or the awareness of increased breathing.
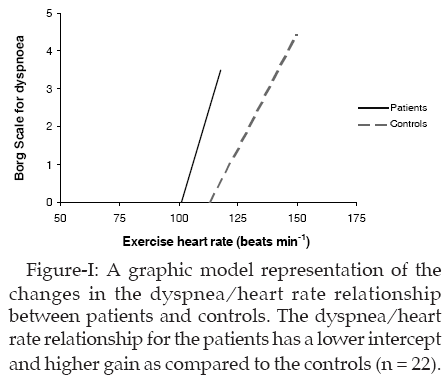
When the participants� Borg scores were plotted against their heart rates, a linear relationship was observed (Figure-1). Linear regression was applied, and from this, the x-axis intercept (beats min-1) and the slope (Borg score min beats-1) were calculated.
A comparison of the patients� and controls� data was performed using these parameters. A paired t-test was used to compare differences and the level of probability taken as significant was 5% (p<0.05).
RESULTS
A. Physical characteristics of study participants:
There were no significant differences in age, BMI or maximum exercise heart rate (Table-I). ST depression was more severe for the subjects with positive ETTs relative to controls (data not shown). Five out of 22 cases (22.7%) were current smokers while 3 (13.6%) were ex-smokers. However in controls, 10 (45.4%) were current smokers and 1 (4.5%) was an ex-smoker.

B. Linearity of the dyspnea/heart rate relationship: The relationship between the Borg score and heart rate was approximately linear (r2 being greater than 0.6 in all relationships) and significant (p < 0.001) for both cases and controls. There was a wide range for the r2 values, slope and intercept for all participants as shown in Table-II. Of note is the fact that the subjects with positive ETT displayed more variation in the slope and intercept of the dyspnea-heart rate relationship as compared to the controls.
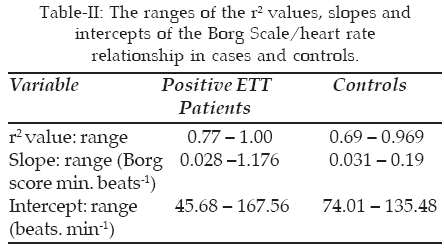
C. Comparison of the intercept and slope of the dyspnea-heart rate relationship between cases and controls: The intercept was found to be significantly lower in cases when compared with controls. The slope was increased in subjects with positive ETT relative to their controls; however, this effect was not statistically significant. The maximum Borg score did not show any significant difference between cases and controls (Table-III).
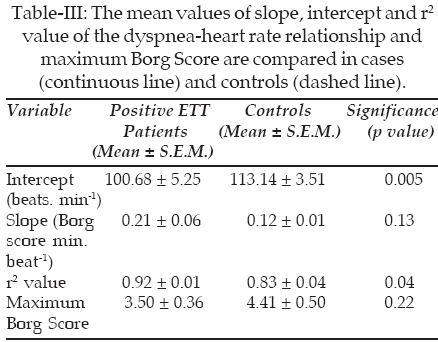
The significant decrease in intercept (threshold) of this relationship indicates a greater level of dyspnea at any given level of heart rate. A graphic representation of the changes in the dyspnea-heart rate relationship between cases and controls is shown in Figure-1.
DISCUSSION
Studies have shown that the relationship between dyspnea and pulmonary ventilation is linear in response to progressive exercise.11-14 However, dyspnea-heart rate relationship has been recently shown to imitate a similar response to that of dyspnea/ventilation relationship.15 This study shows for the first time that dyspnea in cardiac patients can be quantified using the indices of the dyspnea-heart rate relationship in response to a progressive increase in workload.
This study answers the cardinal question whether the dyspnea-heart rate relationship can be investigated in subjects undergoing ETT and what might be the nature of the relationship. Important observations from this study are a) the relationship between dyspnea and heart rate is linear in both subjects who have a positive ETT and those who tested negative, b) the onset of dyspnea is earlier in these patients as evidenced by lower threshold (intercept) of dyspnea-heart rate relationship and c) this relationship can be used during ETT to assess the response of subjects to exercise. The main effect is mediated through the modulation of the threshold, not a change in gain.
Since ETT is a screening tool and it has both a false positive and a false negative rate, our subjects who tested positive cannot be labeled with a diagnosis of Coronary Artery Disease (CAD). Therefore, a diagnosis can only be established after cardiac catheterization shows significant coronary stenosis. Since the dyspnea-heart rate relationship was measured in both ETT positive and negative subjects, it might be reasonable to assume that such a methodology would be useful in screening patients with potential CAD. However, the intercept and the gain as proposed markers of breathlessness have to be analysed in patients with positive ETT and angiography in another study.
Bypass Grafting (Patients of CAD who had Coronary Artery CABG) report a decrease in dyspnea;16 this effect was independent of age and pre-operative Left Ventricular Ejection Fraction (LVEF).6 If we extrapolate our results to CAD patients, it might be suggested that this decrease is mediated via an increase in threshold of the dyspnea-heart rate relationship. Since dyspnea is an important symptom of undiagnosed CAD,3 it would seem that changes in the dyspnea-heart rate relationship might precede the typical EKG changes and chest pain. These are considered to be pathognomonic signs and symptoms of cardiac ischemia.
ETT is regularly employed in clinical practice for screening out patients with high risk of having ischemic heart disease.8 Exertional angina and EKG changes consistent with cardiac ischemia constitute the criteria on the basis of which a given patient is categorized as high risk or low risk. Exertional dyspnea is also an important symptom in such patients. However, this symptom bears no role in the decision whether a test is positive or negative. The obvious reason for this is the fact that exertional dyspnea is also a symptom of normal healthy subjects undergoing exercise.15 Although efforts have been made to distinguish between dyspnea of cardiac and pulmonary origin,17 there is no method to distinguish the pathological exertional dyspnea from non-pathological one and therefore the clinical utility of this symptom in the setting of ETT is very limited. However, this study has highlighted the difference between the two on the basis on intercept and slope variables of the dyspnea-heart rate relationship.
To our knowledge, this is the first study to have dissected out the differences between pathological and non-pathological dyspnea. However, we cannot comment on the origin of this dyspnea since the result of ETT does not give away this information. The exertional dyspnea evaluated in normal controls and patients cannot be discriminated, whether it is cardiac or pulmonary in origin or has a component of both. It may, however, be hypothesized that the dyspnea in healthy controls is completely exertional in nature, whereas that in cases may have an additional component of exercise-induced pulmonary hypertension.18
In conclusion, this study confirms that the exertional dyspnea-heart rate relationship can be quantified in patients with myocardial ischemia. The relationship is linear. A significant decrease in intercept is indicative of higher dyspnea at any given heart rate. The intercept and the gain of this linear relationship can thus be developed as quantifiable potential markers of dyspnea to screen patients with myocardial ischemia before heart attack. Furthermore, these markers might open the avenue for mathematical modeling and understanding the basic mechanisms involved in the genesis of cardiac and pulmonary dyspnea.
ACKNOWLEDGEMENTS
This manuscript is dedicated to the fond memories of Dr Sarwar Zuberi.
The authors thank Dr Khawar Abbas Kazmi, Department of Medicine, Division of Cardiology, Aga Khan University for his contribution in making this study possible. We also thank Taqdees Mariam and Nadir Khan for their assistance in data collection and analysis.
REFERENCES
1. Hagman M, Wilhelmsen L. Relationship between dyspnea and chest pain in ischemic heart disease. Acta Med Scand Suppl 1981; 644: 16-8.
2. Rampulla C, Baiocchi S, Dacosto E, et al. Dyspnea on exercise. Pathophysiologic mechanisms. Chest 1992; 101 (5 suppl): 248s-52s.
3. Cook DG, Shaper AG. Breathlessness, angina pectoris and coronary artery disease. Am J Cardiol 1989; 63 (13): 921-4.
4. Rosengren A, Wilhelmsen L. Respiratory symptoms and long-term risk of death from cardiovascular disease, cancer and other causes in Swedish men. Int J Epidemiol 1998; 27: 962-9.
5. Cook DG, Shaper AG. Breathlessness, lung function and risk of heart attack. Eur Heart J 1988; 9 (11): 1215-22.
6. Herlitz J, Brandrup-Wognesen G, Karlson BW, et al. Limitation of physical activity, dyspnea and chest pain before and two years after coronary artery bypass grafting in relation to preoperative ejection fraction. Scand Cardiovasc J 2000; 34 (1): 65-72.
7. Mahler DA, Horowitz MB. Perception of breathlessness during exercise in patients with respiratory disease. Med Sci Sports Exerc 1994; 26 (9): 1078-81.
8. Hill J, Timmis A. Exercise Tolerance Testing. Br Med J 2002; 324 (7345): 1084-7.
9. Older P, Hall A, Hader R. Cardiopulmonary Exercise Testing as a screening test for preoperative management of major surgery in the elderly. Chest, 1999; 116: 355-62.
10. Adams L, Chronos N, Lane R. The measurement of breathlessness induced in normal subjects: validity of two scaling techniques. Clin Sci (Colch) 1985; 69: 7-16.
11. Mahler DA, Faryniarz K, Lentine T, Measurement of breathlessness during exercise in asthmatics. Predictor variables, reliability and responsiveness. Am Rev Respir Dis 1991; 144 (1): 39-44.
12. Teramoto S, Fukuchi Y, Nagase T. Quantitative assessment of dyspnea during exercise before and after bullectomy for giant bulla. Chest 1992; 105 (5): 1362-66.
13. Noseda A, Carpiaux JP, Schmerber J. Dyspnoea and flow-volume curve during exercise in COPD patients. Eur Respir J 1994; 7 (2): 279-85.
14. Ottanelli R, Rosi E, Romagnoli I. Perception of bronchoconstriction and bronchial hyper-responsiveness in asthma. Clin Sci (Colch) 2000; 98 (6): 681-7.
15. Ahmad HR, Subhan MMF, Bokhari SSI. Intercept shift in the breathlessness/ventilation relationship in response to progressive increase in workload. Adv Exp Med Biol 2001; 499: 383-8.
16. Herlitz J, Karlson BW, Sjoland H. Physical activity, symptoms of chest pain and Dyspnea in patients with ischemic heart disease in relation to age before and two years after coronary artery bypass grafting. J Cardiovasc Surg. 2001; 42 (2): 165-73.
17. Ailani RK, Ravakhah K, DiGiovine B. Dyspnea differentiation index: A new method for the rapid separation of cardiac vs. pulmonary dyspnea. Chest 1999; 116 (4): 100-4.
18. Bossone E, Avelar E, Bach DS. Diagnostic value of resting tricuspid regurgitation velocity and right ventricular ejection flow parameters for the detection of exercise induced pulmonary arterial hypertension. Int J Card Imaging 2000; 16 (6): 429-36.
HOME | SEARCH | CURRENT ISSUE | PAST ISSUES
Professional
Medical Publications
Room No. 522, 5th Floor, Panorama Centre
Building No. 2, P.O. Box 8766, Saddar, Karachi - Pakistan.
Phones : 5688791, 5689285 Fax : 5689860
pjms@pjms.com.pk