|
|
||||
|
Published by : PROFESSIONAL MEDICAL PUBLICATIONS |
||||
|
ISSN 1681-715X |
||||
|
||||
|
- |
||||
|
ORIGINAL ARTICLE |
||||
|
- |
||||
|
Volume 21 |
October December 2005 |
Number 4 |
||
|
|
||||
|
|
||||
Prevalence of celiac disease
among patients with iron
deficiency anemia: Personal experience and review of
literature
Abbas Ali Mansoor1 & Sarkis K. Strak2
ABSTRACT
Background: Iron Deficiency Anemia (IDA) is one of the most common presenting features of Celiac Disease (CD).
Objective: The aim of this study is to find the prevalence of CD among patients with otherwise unexplained IDA and review the literatures.
Setting: Al-Faiha & the Basrah Teaching Hospitals in Basrah for the period from May to December 2004.
Methods: This was a cross sectional study of patients with IDA. Gastroduodenoscopy was performed to determine the cause of anemia. Four biopsies were taken from the second part of duodenum. Small intestinal histologic features were interpreted according to the revised Marsh criteria. Total number of patients was 47, 28 males and 19 females, with a mean age of 26.9±13.3 years.
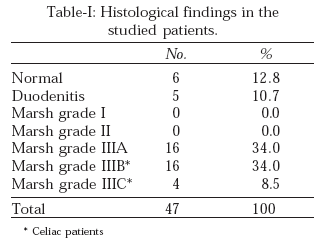
Results: Twenty of 47 patients (42%) had biopsy result consistent with CD. All CD patients were at age of 28 year and below, except one who was 45 year of age. Out of 20 CD patients, 16 were having Marsh grade IIIB and 4 with Marsh grade IIIC. Fourteen were males and 6 were females
Conclusion: All patients with other wise unexplained IDA should have upper gastrointestinal endoscopy with at least four biopsies from the duodenum to exclude CD.
Key words: Celiac Disease (CD), Iron Deficiency Anemia (IDA), duodenal biopsy
Pak J Med Sci October-December 2005 Vol. 21 No. 4 413-416
1. Dr. Abbas Ali Mansour MD
2. Dr. Sarkis K. Strak FRCP
1-2: Department of Medicine,
Basrah College of Medicine,
Basrah, IRAQ
Correspondence:
Dr. Abbas Ali Mansour
Hattin Post Office Box: 142,
Basrah-42002 IRAQ
E-mail: aambaam@yahoo.com
Received for publication June 16, 2005
Accepted for publication September 1, 2005
Introduction
Celiac Disease (CD) is an immune-mediated enteropathy triggered by the ingestion of gluten-containing grains (including wheat, rye, and barley) in genetically susceptible persons.
1 Adult and adolescent patients with CD may present with symptoms in almost any hospital department.2Iron Deficiency Anemia (IDA) is one of the most common presenting features of CD.3 IDA is also common at the diagnosis in patients with CD.4 Iron deficiency with or without anemia, typically refractory to oral iron supplementation, can be the only presenting sign of CD,5 although as many as one half of anemic patients with untreated CD have iron deficiency.6-10
In 2000, The British Society of Gastroenterology proposed that all adult male patients and postmenopausal female patients, without overt blood loss or any other obvious cause of IDA, should undergo upper gastrointestinal endoscopy, including duodenal biopsy, and colonoscopy or barium enema, with or without flexible sigmoidoscopy.11
Screening for CD in patients with common symptoms of irritable bowel syndrome, IDA, unexplained arthritis, and even chronic elevations of aminotransferases is becoming the accepted standard of practice.11-13
The potential advantages of screening for asymptomatic CD include a reduction in risk for enteropathy-associated T-cell lymphoma, a reversal of unrecognized nutritional deficiency states, resolution of mild or ignored intestinal symptoms, avoidance of other auto-immune disorders, and an improvement in general well-being.14 Proximal intestinal biopsies remain the gold standard for diagnosing patients with suspected CD.15 The characteristic histological features of proximal small bowel villous atrophy (either total or subtotal) with associated crypt hyperplasia and intraepithelial lymphocytosis are the hallmark of diagnosing CD. Many gastroenterologists do not take a follow-up biopsy specimen and the cost-effectiveness of this approach has not been demonstrated. Most clinicians also do not undertake formal gluten challenge to show the resultant deterioration of the small intestinal villous architecture. However, gluten challenge should be performed if there is any doubt concerning the correct diagnosis.2
The aim of this study was to find the prevalence of celiac disease among patients with otherwise unexplained IDA and reviewing the literatures.
Patients And Methods
This was a cross sectional study of patients with IDA carried in Al-Faiha and the Basrah Teaching Hospitals in Basrah for the period from May to December 2004. IDA was defined as hemoglobin <12 g/dl, with hypochromic microcytic morphology, normal hemoglobin electrophoresis, and depleted marrow iron stores (done for 40 patients).
Patients were excluded if they had other significant gastrointestinal diseases like history of surgery or history of frank blood loss during the past three months. Background data were collected from all patients, and gastroduod- enoscopy was performed to determine the cause of anemia. Four biopsies were taken from the second part of duodenum. Small intestinal histologic features were interpreted according to the revised Marsh criteria16-19. This scoring system comprises a spectrum of consecutive mucosal abnormalities that can be seen in gluten- sensitive enteropathy. Marsh 0 is described as normal mucosal architecture, without significant intraepithelial lymphocytic infiltration. Marsh I (lymphocytic enteritis) is normal mucosal architecture with a marked infiltration of villous epithelium by lymphocytes; marked is defined as more than 30 lymphocytes per 100 enterocytes. Marsh-II (lymphocytic enteritis with crypt hyperplasia) consists of intraepithelial lymphocytosis and elongation and branching of crypts in which there is an increased proliferation of epithelial cells. Marsh-III comprises intraepithelial lymphocytosis, crypt hyperplasia, and villous atrophy. There are three distinct stages of villous atrophy.18,19 In Marsh IIIA, partial villous atrophy, the villi are blunt and shortened. Arbitrarily, samples classified as partial villous atrophy if the villus-crypt ratio was less than 1:1. In Marsh IIIB, subtotal villous atrophy, villi are clearly atrophic, but still recognizable, and in Marsh IIIC, total villous atrophy, villi are rudimentary or absent, and the mucosa resemble colonic mucosa. Although characteristic histological features of proximal small bowel villous atrophy (either total or subtotal) with associated crypt hyperplasia and intraepithelial lymphocytosis are the hallmark of diagnosing CD,15 only Marsh IIIB and C, were considered as having CD and started gluten free diet.
Total number of patients was 47, 28 males and 19 females with mean age of 26.9±13.3 years.
Results
Twenty (42%) patients had biopsy result consistent with CD (Table-I). All CD patients were younger than 28 year, except one patient who was 45 year of age. Out of 20 CD patients, 16 had Marsh grade IIIB and 4 with Marsh grade IIIC. Fourteen were males and 6 were females. Thirteen male had Marsh grade III B while one male had grade IIIC changes. Three females each had Marsh grade IIIB and grade IIIC changes.
Discussion
Celiac Disease may be a surprisingly frequent cause of IDA, which requires duodenal biopsy to confirm the diagnosis. The finding of endoscopic lesions such as esophagitis and gastritis, that may otherwise explain IDA should not preclude small bowel biopsy and thus delay the discovery of CD8,20,21. Accordingly some recommended screening for CD in patients with unexplained IDA even when there is local cause for bleeding in the gut.22
CD is not an uncommon cause of IDA in patients >65 years and in children below the age of 12 years.23,24 In this study, only one patient with CD was 45 years of age.
To increase the diagnostic yield of CD we have obtained 4 biopsies from the second part of the duodenum, since histological changes can be patchy15. However some patients with CD had biopsies which are not consistent with CD, the so-called potential CD. These patients might show immunologic abnormalities characteristic for the disorder (e.g., positive IgA or IgG to endomysium, and IgA or IgG tissue transglutaminase antibodies, now recognized as the offending endogenous antigen in CD)25, a "celiac intestinal antibody pattern," and increased intraepithelial lymphocytes.26 The reason why both IgA and IgG antibodies are used in the diagnosis of CD, is that those people are at least five times more frequently IgA deficient than healthy control subjects, and appropriate screening for CD requires measurement of endomyseal or tissue transglutaminase antibodies and IgA.27 Unfortunately these autoantibodies were not available at the time of this study was conducted to aid the diagnosis in those patients with negative histology.
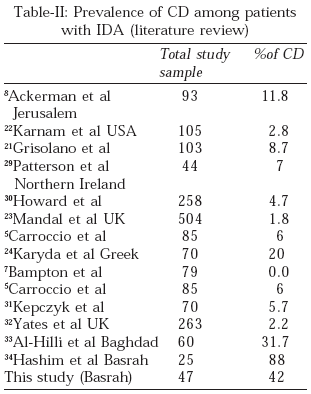
The prevalence of CD in this study was 42% compared to other studies (Table-II). The differences between studies may be due to various factors, including age, disease prevalence in the country of origin, and referral bias, 8-24 together with different diagnostic tools used for the diagnosis. For instances in some centers the presence of a few inflammatory cells in an otherwise normal epithelium is sufficient to make the diagnosis in a seropositive patient.17, 19, 28
Whilst the classic presentation of CD with diarrhoea and malabsorption has become relatively rare (prevalence from 1 in 2000 to 1 in 10 000 in the west), atypical, oligosymptomatic, or even asymptomatic manifestations (with patients having villous flattening of various degrees) are frequent, with an estimated prevalence of 1 in 200 in Europe and the USA, making CD one of the most common inherited disorders. Untreated patients with oligosymptomatic like IDA or asymptomatic CD are of concern, since they might have an increased risk of disease exacerbation later, secondary autoimmune disorders, and gastrointestinal or haematological cancers.1,2
In conclusion, all patients with other wise unexplained IDA should have endoscopy with at least four biopsies from the duodenum to exclude CD.
References
1. Schuppan D. Novel concepts of celiac disease pathogenesis. Gastroenterol 2000; 119: 234–42
2. Ciclitira PJ, King AL, Fraser JS. AGA technical review on celiac sprue. American Gastroenterological Association. Gastroenterol 2001; 120:1526–40
3. Hin H, Bird G, Fisher P, et al. Coeliac disease in primary care: case finding study. BMJ 1999; 318:164–7
4. Cluysenaer OJJ, vanTon gerenJHM. Malabsorp- tion in celiac sprue. The Hague: Martinus Nijhoff, 1977.
5. Carroccio A, Iannitto E, Cavataio F, Montalto G, Tumminello M, Campagna P et al. Sideropenic anemia and celiac disease: one study, two points of view. Dig Dis Sci 1998;43:673–8
6. Hoffbrand AV. Anemia in adult celiac disease. Clin Gastroenterol 1974;3:71-89
7. Bampton PA, Holloway RH. A prospective study of the gastroenterological causes of iron deficiency anaemia in a general hospital. Aust NZ J Med 1996;26:793-9
8. Ackerman Z, Eliakim R, Stalnikowicz R, Rachmilewitz D. Small bowel biopsy in the endoscopic evaluation of adults with iron deficiency anemia. Am J Gastroenterol 1996; 91:2099-2102
9. McIntyre AS, Long RG. Prospective survey of investigations in out-patients referred with iron deficiency anemia. Gut 1993;34:1102–7
10. Corazza GR, Valentini RA, Andreani ML, D’Anchino M, Leva MT, Ginaldi L et al. Subclinical celiac disease is a frequent cause of iron-deficiency anaemia. Scand J Gastroenterol 1995; 30:153-6
11. Goddard AF, McIntyre AS, Scott BB. Guidelines for the management of iron deficiency anaemia. British Society of Gastroenterology. Gut 2000; 46(IV):1–5
12. Treem WR. Emerging concepts in celiac disease. Curr Opin Pediatr 2004; 16:552-9
13. Bardella MT, Vecchi M, Conte D, Del Ninno E, Fraquelli M, Pacchetti S et al. Chronic unexplained hypertransaminasemia may be caused by occult celiac disease. Hepatology 1999; 29:654–7
14. Corrao G, Corazza GR, Andreani ML, et al. Serological screening for celiac disease: Choosing the optimal procedure according to various prevalence values. Gut 1994; 35:771
15. Frenz MB, Mee AS.Making the diagnosis of coeliac disease: is there a role for push enteroscopy? Eur J Gastroenterol Hepatol 2004, 16:1127–9
16. Marsh MN. The immunopathology of the small intestinal reaction in gluten sensitivity. Immunol Invest. 1989;18:509-31
17. Marsh MN. Gluten, major histocompatibility complex and the small intestine: a molecular and immunobiologic approach to the spectrum of gluten sensitivity ("celiac sprue"). Gastroenterology 1992;102:330-54
18. Wahab PJ, Crusius JBA, Meijer JWR, et al. Gluten challenge in borderline gluten sensitive enteropathy. Am J Gastroenterol 2001;96:1464-9
19. Oberhuber G, Granditsch G, Vogelsang H. The histopathology of celiac disease: time for a standardized report scheme for pathologists. Eur J Gastroenterol Hepatol 1999; 11:1185–94
20. Ackerman, Z, Eliakim, R, Stalnikowica, R, Rachmilewitz, D. Role of small bowel biopsy in the endoscopic evaluation of adults with iron deficiency anemia. Am J Gastroenterol 1996; 91: 2099
21. Grisolano SW, Oxentenko AS, Murray JA, Burgart LJ, Dierkhising RA, Alexander JA. The Usefulness of Routine Small Bowel Biopsies in Evaluation of Iron Deficiency Anemia. J Clin Gastroenterol 2004; 38:756-60
22. Karnam US, Felder LR, Raskin JB. Prevalence of occult celiac disease in patients with iron-deficiency anemia: a prospective study.South Med J 2004; 97:30-4
23. Mandal AK, Mehdi I, Munshi SK, Lo TC. Value of routine duodenal biopsy in diagnosing coeliac disease in patients with iron deficiency anaemia. Postgrad Med J 2004; 80:475-7
24. Karyda S, Mouskou S, Avgoustidou P et al. The clinical spectrum of celiac disease in Greek children –a single center experience. J Pediatr Gatroenterol Nutr 2004; 39:s222 (Abstract)
25. Shan L, Molberg O, Parrot I, Hausch F, Filiz F, Gray GM et al. Structural basis for gluten intolerance in celiac sprue. Science 2002; 297:2275–9
26. Arranz, E, Ferguson, A. Intestinal antibody pattern of celiac disease: Occurrence in patients with normal jejunal biopsy histology. Gastroenterol 1993; 104:1263
27. Farrell RJ, Kelly CP. Diagnosis of celiac sprue. Am J Gastroenterol 2001; 96:3237–46
28. Marsh M, Crowe P: Morphology of the mucosal lesion in gluten sensitivity. Bailliere’s Clinical Gastroenterol 1995; 9:273–93
29. Patterson RN, Johnston SD. Iron deficiency anaemia: are the British Society of Gastroenterology guidelines being adhered to? Postgrad Med J 2003; 79:226-8
30. Howard MR, Turnbull AJ, Morley P, Hollier P, Webb R, Clarke A. A prospective study of the prevalence of undiagnosed celiac disease in laboratory defined iron and folate deficiency. J Clin Pathol 2002; 55:754-7
31. Kepczyk T, Kadakia SC. Prospective evaluation of gastrointestinal tract in patients with iron-deficiency anemia. Dig Dis Sci 1995; 40:1283–9
32. Yates JM, Logan ECM, Stewart RM. Iron deficiency anaemia in general practice: clinical outcomes over three years and factors influencing diagnostic investigations. Postgrad Med J 2004; 80:405–10
33. Al–Hilli H, Salmo N, Jaber A. Iron deficiency anemia as the presenting features of celiac disease. Proceeding of the thirty seventh Iraqi medical conference. Baghdad 2002. Abstract book p-16
34. Hashim AR, Abdulwahid R. Iron deficiency anemia as a sole presentation of gluten sensitive enteropathy. Med J Basrah University 2003; 21:120-24
HOME | SEARCH | CURRENT ISSUE | PAST ISSUES
Professional
Medical Publications
Room No. 522, 5th Floor, Panorama Centre
Building No. 2, P.O. Box 8766, Saddar, Karachi - Pakistan.
Phones : 5688791, 5689285 Fax : 5689860
pjms@pjms.com.pk
