|
|
||||
|
Published by : PROFESSIONAL MEDICAL PUBLICATIONS |
||||
|
ISSN 1681-715X |
||||
|
||||
|
- |
||||
|
ORIGINAL ARTICLE |
||||
|
- |
||||
|
Volume 24 |
April - June 2008 (Part-I) |
Number 2 |
||
|
|
||||
|
||||
|
|
||||
|
Published by : PROFESSIONAL MEDICAL PUBLICATIONS |
||||
|
ISSN 1681-715X |
||||
|
||||
|
- |
||||
|
ORIGINAL ARTICLE |
||||
|
- |
||||
|
Volume 24 |
April - June 2008 (Part-I) |
Number 2 |
||
|
|
||||
|
||||
Effect of pregnancy on total antioxidant
capacity in Nigerian women
Idogun ES1, Odiegwu ME2, Momoh SM3, Okonofua FE4
ABSTRACT
Objective: To assess the total antioxidant capacity during the 3rd trimester of normal pregnancy with the aim of establishing the status of total antioxidant capacity (TAC) in pregnant Nigerians.
Methodology: The subjects consisted of 20 pregnant Nigerian women and 20 non-pregnant women. The cases were age matched with the controls. Total antioxidant capacity was assayed in both the cases and controls using standard methods of total antioxidant capacity assay.
Results: The total antioxidant capacity was lower in the pregnant women 0.84mmol/L compared with the controls 1.00mmol/L, P = 0.007 ‘t’ test.
Conclusion: We observed low total antioxidant capacity in Nigerian women with normal pregnancy. This preliminary report, calls for further investigations.
KEY WORDS: Pregnancy, Oxidative stress, Antioxidant capacity, Nigerian women.
Pak J Med Sci April - June 2008 (Part-I) Vol. 24 No. 2 292-295
1. Idogun ES,
Department of Chemical Pathology,
College of Medicine, University of Benin.
Nigeria.
2. Odiegwu ME,
3. Momoh SM,
4. Okonofua FE,
2-4: Department of Obstetrics and Gynecology,
College of Medicine, University of Benin
Benin City, Nigeria.
Correspondence
Dr. E.S Idogun,
Department of Chemical Pathology,
University of Benin Teaching Hospital,
P.M.B 1111, Benin – City,
Nigeria.
E-mail:
sylvesteridogun@yahoo.co.uk
* Received for Publication: July 10, 2007
* Revision Received: January 17, 2008
* Revision Accepted: January 20, 2008
INTRODUCTION
Oxidative stress refers to the excessive generation of oxidants in the body. Pregnancy is a stressful condition; we hypothesized that it could lead to increased levels of oxidants and therefore reduce total antioxidant capacity (TAC). Further the effect of pregnancy on TAC may be different in Caucasians women from Nigerian women because of differences in diet and increased stressful environment in which Nigerians become pregnant.
Pregnancy is a physiological process, yet many diseases occur for the first time during pregnancy
1 because of oxidative stress. Pregnancy therefore tends to promote oxidative stress2 as markers of oxidative stress are known to be raised in normal pregnancy.3 Total antioxidant capacity represents the balance between oxidative stress (oxidants) and the neutralizing systems (antioxidants). The oxidants are mainly reactive oxygen species and their derivatives e.g. peroxynitrite anion,4 In contrast, homeostasis against the effects of reactive oxygen species and their derivatives is maintained by antioxidants such as catalase, super oxide dismutase, beta-carotene, vitamin C, vitamin E, glutathione peroxides, ceruloplasmin and transfer in. These agents intercept, modify or destroy the reactive free radicals.5 Antioxidants therefore mitigate oxidative stress in pregnancy. Total antioxidant capacity is a measure of the neutralizing effects of antioxidants.In Nigeria, there is paucity of literature on the antioxidant/oxidative stress in pregnancy. The objective of this study therefore is to assess the total antioxidant capacity during the 3
rd trimester of normal pregnancy with the aim of establishing the status of total antioxidant capacity (TAC) in pregnant Nigerians.METHODOLOGY
Subjects: Twenty pregnant women were randomly selected from among Antenatal clinic attendants of the University of Benin Teaching Hospital. The women were aged between 20 years and 39 years, in the third trimester of pregnancy, and without any obvious complications of pregnancy. They were not diabetic or hypertensive and had no known medical complications of pregnancy. The controls were non-pregnant women. They were age-matched (1:1) with the cases. They were examined physically and confirmed not to be pregnant by examination of early morning urine for beta-hCG and uterine ultrasound. Both cases and controls were sampled with questionnaire that documented their ages, education status, occupation, gestational age, and drug history. All the participants gave informed consent after the study was approved by the hospital ethical committee.
Laboratory Assay: Ten millitres of venous blood were collected from the ante-cubital veins of the participants, into lithium heparin specimen bottles, after routine aseptic procedure. The plasma was separated after centrifugation with clean Pasteur pipettes. Total antioxidant status (TAS) was analyzed on the same day of sample collection. The remaining samples were stored frozen for other analyses. TAS was measured using TAS kit manufactured by Randox laboratory; U.S.A It was by a photometric quantitative measurement,
6 using a conventional spectrophotometer after color development.Statistical Analysis: All data were analyzed by computer using Instate graph pad Soft ware. The mean, SD, SEM were calculated. Student ‘t’-test was applied to test for significance at 95% confidence interval.
RESULTS
The study group was made of 20 pregnant women, and 20 non-pregnant age- matched controls. The mean age of the patients was 31.25 ± 3.24 years, 95% CI 29.733 to 32.77. This was not different from the mean age of the controls, 30.9 ± 2.9, P= 0.4 t-test. The mean gestational age of the cases was 33 ± 5.58 weeks, 95% CI 30.54 to 35.76. The educational status was not significantly different in controls and the cases, P = 0.71 (chi square test). 75% of the cases had tertiary education, 20% secondary education and 5% primary education. A total of 75% of them were gainfully employed while 25% had no employment. The total antioxidant capacity was lower in pregnant women 0.842 ± 0.1711mmol/L compared with the controls 1.003 ± 0.187mmol/L, P = 0.0072 (Student‘t’ test), Table I.
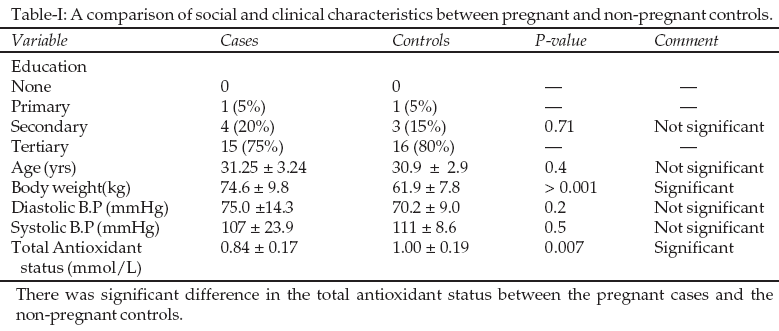
DISCUSSION
Pregnancy, though a physiological process promotes oxidative stress.
2,3 Antioxidants are used as defense mechanisms against oxidative stress. Total antioxidant capacity (TAC) represents the balance between oxidative stress and the neutralizing systems. In our study, we found low level of total antioxidant capacity in pregnant women with normal pregnancy. Although most other studies report low levels of specific antioxidants in pregnancy.2,3 Our study confirmed a global reduction in total antioxidant capacity during normal pregnancy.Oxidative processes are not entirely harmful in pregnancy. They exert a fundamental regulatory function during pregnancy. At the beginning of pregnancy, when a certain grade of inflammatory change is necessary to the trophoblast invasion of maternal tissue, the activation of the process by nitric oxide and reactive oxygen species is welcome. Indeed these products modulate the metalloproteinases, which are responsible for the remodeling of uterine extracellular matrix.
7 Problems arise when there is an imbalance between the pro-oxidants and antioxidants either because of excessive generation of pro-oxidants or because of reduced levels of the antioxidant systems, leading to inadequate reducing capacity of the antioxidants.8 The consequence of this, is an increased plasma levels of pro-oxidant free radicals (e.g. hydroxyl radical (HO.), superoxide anion radical (02–), nitric oxide (NO.), hydrogen perioxide (H202), hypochlorous acid (HOCl) and peroxynitrite anion (ONOO–). These free radicals attack polyunsaturated fatty acids or cholesterol in membranes or lipoproteins (lipid hydroperoxidation). Lipid hydroperoxides function in normal physiology by regulating enzymes and redox sensitive genes.9,10 However this necessary physiology in normal pregnancy can become pathologic if lipid peroxidation becomes uncontrolled resulting in cellular dysfunction and damage and increase in vascular resistance, as in preeclampsia.11Of particular documented evidence is the subliminal shortage of selenium and vitamin E which may be uncovered under the physiological stress of pregnancy, when mother and conceptus both drawn on a limited supply of this trace element and vitamin. These deficiencies will lead to a relative functional deficiency of thioredoxin reductase, a system that is needed for normal function of RNA and DNA.
8 These deficiencies will also increase oxidative stress of pregnancy that may lead to preeclampsia and eclampsia.8 Oxidative stress therefore will lead to increased utilization of antioxidant system and a low level of total antioxidant capacity. This explains why we found low TAC in our pregnant women.The consequences of reduced antioxidant systems in pregnancy are many. Reduced antioxidants may be a cause of reduced placental efficiency and calcification.
12 It could also be a cause of fetal malformation, pregnancy complications such as preeclampsia and eclampsia as well as aggravate obstructive airway disease13 and gestational diabetes.We may therefore infer from our study that most of the complications we see among our pregnant women may be related to low levels of TAC. Recently Onah etal
14 reported a profile of the major causes of maternal mortality in Eastern Nigeria. Preeclampsia/eclampsia was responsible for about 16% cause of maternal death. Preeclampsia and elcampsia have been linked to imbalance in the antioxidant systems.8 A pilot study in London women shows that vitamins C and E given antenatally in high dosages in pregnancy substantially lowered the incidence of preeclampsia.15 In China, the tendency to hypertension in pregnancy has been relieved by adding trace amounts of selenium to the diet.16 In Wales, women miscarrying their pregnancy have been shown to have low selenium concentrations.17 In Enugu Nigeria, Uchenna etal 3 reported specifically on vitamins C and E to be reduced in pregnant women.We therefore suggest that dietary supplementation with antioxidants such as vitamin C, E and selenium and other antioxidants will improve the total antioxidant status and reduce pregnancy complications, in our pregnant women. This will be most beneficial in our community where quality obstetric care is limited to the metropolitan areas, and obstetric intervention is poor in rural areas. We hypothesize that dietary or supplemental antioxidants during pregnancy will improve placental function and fetal well-being, reduction in the incidence of fetal malformation, and placenta calcification. It reduces the incidence and severity of vascular resistance diseases in pregnancy such as pre-eclampsia and eclampsia and gestational diabetes.
Our findings though a preliminary support for antioxidant supplements in pregnancy, intervention and prospective studies with specific antioxidants are necessary in this direction.
CONCLUSIONS
We report a low level of total antioxidant capacity in the third trimester of normal pregnancy. However the use of exogenous antioxidant supplements, during pregnancy in the prevention of pregnancy complications require more detailed clinical studies.
ACKNOWLEDGMENT
The Women’s Health and Action Research Centre (WHARC) provided funding for this study.
REFERENCES
1. Kaja RJ, Greer LA. Manifestations of chronic disease during pregnancy. JAMA 2005;294(21):2751-7.
2. Uchenna IN, Fidelis EE. Serum ascorbic acid levels during pregnancy in Enugu, Nigeria. J Coll Med 2005;10(1):43-5.
3. Jonathan MM, Nitin KG, Marit JR, Knight M, Elizabeth AL, Sumwan D, et al. Circulating markers of oxidative stress is raised in normal pregnancy and preeclampsia. Briti J Obs Gyn 1998;105:1195-9.
4. Darley-Usmar V, Halliwel B. Blood radicals: Reactive nitrogen species, reactive oxygen species, transitional metal ions and vascular system. Pharm Res 1997;13:649-62.
5. Halliwell B, Guttertidge JMC. Antioxidant Defences ch.3 in Free Radicals in Biology and Medicine Oxford: University Press 1999.
6. Miller NJ, Rice-Evans C, Davies MJ, Gopinathan V, Miller A. Clinical Science 1993;84:407-12.
7. Biondi C, Paran B, Langhi L, Fiorini S, Vesce F. The role and modulation of the oxidative balance in pregnancy. Curr Pharm Des 2005;11(16):2075-89.
8. Stark JM. Inadequate reducing system in pre-eclampsia: a complementary role for vitamin C and E with thioredoxin-related activities. Briti J Obs Gyn 2001;108:339-43.
9. Smith WL, Marnett LJ, DeWitt DL. Prostaglandin and thromboxane biosynthesis. Pharmacol Ther 1991;49:153-79.
10. Sen CK, Packer L. Antioxidant and redox regulation of gene transcription. FASEB J 1996;10:709-20.
11. Carl AH. Oxidative stress in the pathogenesis of preeclampsia. http://www.ebmonline.org/cgi/content/full. Accessed on January 10, 2008.
12. Klesgee LM, Murray DM, Brow JE, Cliver SP, Goldenberg RL. Relations of cigarette smoking and dietary antioxidants with placenta calcification. Amer J Epidemiology 1998;147:127-35.
13. Hug Cassino PA. Antioxidant nutrients and pulmonary function: The third National and Nutrition Examination Survey (NHAMES III). Am J Epidol 2000;151(10):975-81.
14. Onah HE, Okaro JM, Umeh U, Chigbu CO. Maternal morality in health institutions with emergency Obstetric care facilities in Enugu state, Nigeria. J Obs Gyn 2005;25(6):569-74.
15. Happell LC, Seed PT, Briley AL. Effect of antioxidants on the occurrence of pre-eclampsia in women at high risk. Lancet 1999;354:810-15.
16. Lu B, Zhang SW, Huang B, Liu W, Li CF, Changes in selenium in patients with pregnancy induced hypertension, Chin J Obstet Gynaecol 1990;25:325-7.
17. Barrington JW, Lindsay P, James D, Smith S, Roberts A. Selenium deficiency and miscarriage: A possible link? British J Obstet Gynaecol 1996;103:130-2.
HOME | SEARCH | CURRENT ISSUE | PAST ISSUES
Professional
Medical Publications
Room No. 522, 5th Floor, Panorama Centre
Building No. 2, P.O. Box 8766, Saddar, Karachi - Pakistan.
Phones : 5688791, 5689285 Fax : 5689860
pjms@pjms.com.pk