|
|
||||
|
Published by : PROFESSIONAL MEDICAL PUBLICATIONS |
||||
|
ISSN 1681-715X |
||||
|
||||
|
- |
||||
|
ORIGINAL ARTICLE |
||||
|
- |
||||
|
Volume 24 |
July - September 2008 |
Number 4 |
||
|
|
||||
|
|
||||
|
|
||||
|
Published by : PROFESSIONAL MEDICAL PUBLICATIONS |
||||
|
ISSN 1681-715X |
||||
|
||||
|
- |
||||
|
ORIGINAL ARTICLE |
||||
|
- |
||||
|
Volume 24 |
July - September 2008 |
Number 4 |
||
|
|
||||
|
|
||||
activity of certain drugs in CombinationIn vitro
Ghaleb Mohammad Adwan1, Bassam Ali Abu-Shanab2,
Kamel Mohammad Adwan3
ABSTRACT
Objectives: This study has been done to evaluate the interaction between ethanolic extracts of Rhus coriaria (seed), Psidium guajava (Leaf), Lawsonia inermis (Leaf) and Sacropoterium spinosum (seed) and antimicrobial drugs including oxytetracycline HCl, enrofloxacin, gentamicin sulphate and sulfadimethoxine against four clinical isolates of methicillin-resistant Staphylococcus aureus (MRSA).
Methodology: Evaluation of the interaction between ethanolic extracts and different antimicrobial agents has been done using well-diffusion method.
Results: It showed that ethanolic extracts increase the inhibition zones of oxytetracycline HCl, gentamicin sulphate, and sulfadimethoxine, while combinations between these plant extracts and enrofloxacin decrease inhibition zone.
Conclusion: This study probably suggests the possibility of concurrent use of these antimicrobial drugs and plant extracts in combination in treating infections caused by S. aureus strains or at least the concomitant administration may not impair the antimicrobial activity of these antibiotics.
KEYWORDS: Ethanolic plant extracts, Antibiotics, Drugs, Synergism, Antagonism, Staphylococcus aureus, MRSA.
Pak J Med Sci July - September 2008 Vol. 24 No. 4 541-544
How to cite this article:
Adwan GM, Abu-Shanab BA, Adwan KM.In vitro activity of certain drugs in Combination with plant extracts against Staphylococcus aureus infections. Pak J Med Sci 2008;24(4):541-4.
1. Ghaleb Mohammad Adwan,
Department of Biology and Biotechnology,
An-Najah N. University,
P. O. Box (7)-Nablus,
Palestine.
2. Bassam Ali Abu-Shanab,
Faculty of Veterinary Medicine,
An-Najah N. University,
Nablus, Palestine.
3. Kamel Mohammad Adwan,
Department of Biology and Biotechnology,
An-Najah N. University,
P. O. Box (7)-Nablus, Palestine.
Correspondence
Ghaleb Mohammad Adwan,
E-mail: adwang@najah.edu
* Received for Publication: November 24, 2007
* Revision Received: May 14, 2008
* Revision Accepted: May 22, 2008
INTRODUCTION
Infectious diseases still represent an important cause of morbidity and mortality among humans, especially in developing countries. Even though pharmaceutical companies have produced a number of new antibacterial drugs in the last years, resistance to these drugs by bacteria has increased and has now become a global concern. In general, bacteria have the genetic ability to transmit and acquire resistance to drugs used as therapeutic agents.
1 Staphylococcus aureus is recognized as one of the major causes of infections in humans occurring in both the community and the hospital. Methicillin-resistant and multidrug resistant staphylococci have become a major nosocomial pathogen.2 Therefore the importance of identifying new effective antimicrobial agents cannot be overemphasized. Among the potential sources of new agents, medicinal plants have long been investigated. In rational drug therapy, the concurrent administration of two or more drugs is often essential and sometimes mandatory in order to achieve the desired therapeutic goal or to treat co-existing diseases. However, the drug interaction may have different effects on the host as well as the infecting microorganism. The potential benefits of using combined antimicrobial therapy can be treatment of mixed infections, therapy of severe infections in which a specific causative organism is known, enhancement of antibacterial activity, reducing the time needed for long-term antimicrobial therapy and prevention of the emergence of resistant microorganisms.3,4Drug synergism between known antimicrobial agents and bioactive plant extracts is a novel concept and has been recently reported.
1,5-14 Three of the plants used in this study are medicinal plants (Rhus coriaria, Psidium guajava, and Lawsonia inermis) and the fourth (Sacropoterium spinosum) which is considered as animal food. In this in vitro study, we attempted to evaluate the possible antimicrobial interaction between ethanolic extracts of these plants and certain known antimicrobial drugs (oxytetracycline HCl, enrofloxacin, gentamicin sulphate and sulfadimethoxine) against four strains of methicillin-resistant S. aureus (MRSA) using the well diffusion method.METHODOLOGY
Plant material and extract preparation: The plant materials used in this study consisted of R. coriaria (seed), P. guajava (Leaf), L. inermis (Leaf) and S. spinosum (seed), which are growing in Palestine. Ethanolic extracts were prepared as described previously.
15-17 Plant materials were dried in an open air protected from direct exposure to sunlight and 30g of dried plant materials were separately powdered and extracted with 80% ethanol; the extracts were filtered through Whatman No. two filter paper under suction. Extracts were concentrated to dryness in vacuum. Then, 100mg of the dry residue was dissolved in one ml of sterile distilled water.Bacterial strains: Four methicillin-resistant S. aureus (MRSA) strains were isolated from clinical specimens (urine and semen). These isolates were identified as S. aureus according to colonial and microscopic morphology, growth on Mannitol Salt agar, 5% blood sheep agar, positive catalase, and coagulase production. Methicillin resistance was carried out in the microbiology laboratories of An-Najah National University, Palestine, using the disk diffusion method.
18 Methicillin (5µg) disks (Oxoid) were used, and inhibition zones were determined in accordance with procedures of the National Committee for Clinical Laboratory Standard (NCCLS).19 A reference strain [Bacillus subtilis ATCC6633] was also tested.Antimicrobial drugs: Four drugs were evaluated for synergism assays. These included oxytetracycline HCl (10%), enrofloxacin (10%), gentamicin sulphate (50%) and sulfadimethoxine as sodium (40%). All these antimicrobial agents were produced by Jerusalem Pharmaceutical CO. Balsam Branch, and were diluted to a final concentration 100µg/ml.
Antimicrobial tests: Antibacterial activity was measured using a well diffusion method according to the National Committee for Clinical Laboratory Standard.
19 Briefly, Petri plates containing 20ml of Mueller Hinton agar medium were inoculated with a 24 hour culture of the bacterial strains. Wells (6mm diameter) were punched in the agar and filled with 30ěl of plant extracts or antibiotics and in case of combination 30µl of each has been added into well. Triplicates of each plate have been done. The plates were incubated at 37°C for 24 hour. The antibacterial activity was assessed by measuring the diameter of the area in which bacterial growth was inhibited around the well. The average of three replicates for each extract, antibiotic and combination were calculated.RESULTS
Antimicrobial mechanisms of the drugs used here were variable and the nucleic acid synthesis inhibitor did not show synergistic effect. However, competitive inhibitor (sulfadimethoxine as sodium) and protein synthesis inhibitors (oxytetracycline HCl, gentamicin sulphate) showed strongest synergistic interaction. The data pertaining to the antimicrobial potential of the plant extracts and antibacterial drug combination against S. aureus using well diffusion method are presented in Table-I.
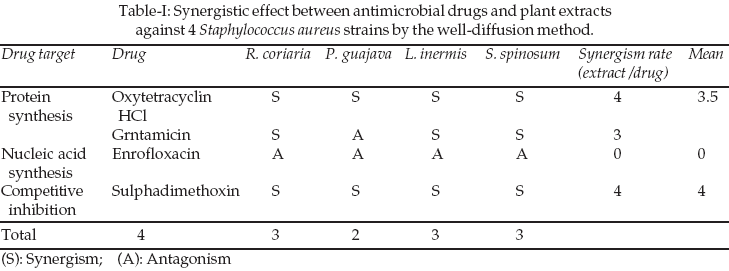
DISCUSSION
Antimicrobial drugs effective for treatment of patients infected with MRSA are limited. Thus, it is important and valuable to find compounds that potentiate antimicrobial activity of antibiotics on MRSA. Our results in this report indicated that all the extracts of the plants studied showed an increase in the antimicrobial activity of certain drugs that can be used against S. aureus, and synergistic interaction of plant extracts is possible with antimicrobial drugs. These results are consistent with previous reports which showed that some plant extracts can increase the activity of antimicrobial drugs in vitro against bacteria.
1,8-12Competitive inhibitor (sulfa drugs as sulfadimethoxine) and protein synthesis inhibitors (aminoglycosides as gentamicin sulphate and tetracyclines as oxytetracycline HCl) showed high synergism rate with plant extracts, while nucleic acid synthesis inhibitor (quinolones as enrofloxacin) did not show this effect. These results are in agreement with that reported previously, which showed that protein synthesis inhibitors showed strongest synergistic interaction and nucleic acid synthesis inhibitor showed weak synergism with plant extracts.
8,9 This high synergism rate shows the need for more studies concerning the molecular basis of these interactions to understand the synergistic mechanism which is fundamental to development of pharmacological agents to treat bacterial infections using medicinal plants.Here we recommended the evaluation of the exact drug-plant ratio at which the interaction in maximal between the plant extract and antimicrobial drug. A wider study with increase in the number of drugs in each group, increased number of clinical isolates, and the identification of the effective compounds in the crude extract are also necessary. It may be deduced that numerous compounds within the crude extract may have interfered with the actions of one another. Once they were separated by various methods, the inhibiting effect of one on the other can be reduced significantly.
CONCLUSION
The results presented in this report were encouraging, although clinical controlled studies are needed to define the real efficacy and possible toxic effects in vivo. This study probably suggests the possibility of concurrent use of these antimicrobial drugs and extracts in combination in treating infections caused by S. aureus strains or at least the concomitant administration of these plants and antimicrobial drugs may not impair the antimicrobial activity of these antibiotics.
REFERENCES
1. Nascimento GGF, Locatelli J, Freitas PC, Silva GL. Antibacterial activity of plant extracts and phytochemicals on antibiotic-resistant bacteria. Braz J Microbiol 2000;31:247-56.
2. National Nosocomial Infections Surveillance (NNIS) System Report, data summary from January 1992 through June 2004, issued October 2004. Am J Infect Control 2004;32:470-85.
3. Hugo WB, Russel AD. Pharmaceutical Microbiology. 1993. Blackwell Scientific Pub., New York.
4. Levinson W, Jawetz E. Medical microbiology and immunology: Examination and board review. 2002. International. 7th ed., Lange Medical Books/McGraw-Hill, New York.
5. Aburjai T, Darwish RM, Al-Khalil S, Mahgzah A, Al-Abbdi A. Screening of antibiotic resistant inhibitors from local plant materials against two different strains of Pseudomonas aeruginosa. J Ethnopharmacol 2001;76:39-44.
6. Shimizu M, Shiota S, Mizushima T, Ito H, Hatano T, Yoshida T, et al. Marked potentiation of activity of beta-lactams against methicillin-resistant Staphylococcus aureus by corilagin. Antimicrob Agents Chemother 2001;45:3198-201.
7. Aqil F, Khan MSA, Owais M, Ahmad I. Effect of certain bioactive plant extracts on clinical isolates of ß-lactamase producing methicilin resistant Staphylococcus aureus. J Basic Microbiol 2005;45:106-14.
8. Junior AF, Balestrin EC, Betoni JE, Orsi Rde O, da Cunha Mde L, Montelli AC. Propolis: anti-Staphylococcus aureus activity and synergism with antimicrobial drugs. Mem Inst Oswaldo Cruz 2005;100:563-6.
9. Betoni JE, Mantovani RP, Barbosa LN, Di Stasi LC, Junior AF. Synergism between plant extract and antimicrobial drugs used on Staphylococcus aureus diseases. Mem Inst Oswaldo Cruz 2006;101:387-90.
10. Esimone CO, Iroha IR, Ibezim EC, Okeh CO, Okpana EM. In vitro evaluation of the interaction between tea extracts and penicillin G against staphylococcus aureus. Afr J Biotechnol 2006;5:1082-6.
11. Chang PC, Li HY, Tang HJ, Liu JW, Wang JJ, Chuang YC. In vitro synergy of baicalein and gentamicin against vancomycin-resistant Enterococcus. J Microbiol Immunol Infect 2007;40:56-61.
12. Horiuchi K, Shiota S, Kuroda T, Hatano T, Yoshida T, Tsuchiya T. Potentiation of antimicrobial activity of aminoglycosides by carnosol from Salvia officinalis. Biol Pharm Bull 2007;30:287-90.
13. Ibezim EC, Esimone CO, Nnamani PO, Onyishi IV, Brown SA, Obodo CE. In vitro study of the interaction between some fluoroquinolones and extracts of kola nitida seed. Afr J Biotechnol 2006;5:1781-4.
14. Ali NH, Kazmi SU, Faizi S. Activity of synergistic combination Amoxy-cassia against Salmonella. Pak J Pharm Sci 2007;20:140-5.
15. Abu-Shanab B, Adwan G, Abu-Safiya D, Adwan G, Abu-Shanab M. Antibacterial activity of Rhus coriaria. L extracts growing in Palestine. J Islamic Univ Gaza (Natural Sciences series) 2005;13:147-53.
16. Abu-Shanab B, Adwan G, Abu-Safiya D, Jarrar N. Antibacterial activities of some plant extracts utilized in popular medicine in Palestine. Turk J Biol 2004;28:99-102.
17. Adwan G, Abu-Shanab B, Adwan K, Abu-Shanab F. Antibacterial Effects of nutraceutical plants growing in Palestine on Pseudomonas aeruginosa. Turk J Biol 2006;30:239-42.
18. Bauer AW, Kirby WMM, Sherris JC, Truck M. Antibiotic susceptibility testing by a standardized single disc method. Am J Clin Pathol 1966;45:493-6.
19. National Committee for Clinical Laboratory Standards (NCCLS). Performance standards for antimicrobial disk and dilution susceptibility tests for bacteria isolated from animals. Pennsylvania, 1999. USA, (M31-A).
HOME | SEARCH | CURRENT ISSUE | PAST ISSUES
Professional
Medical Publications
Room No. 522, 5th Floor, Panorama Centre
Building No. 2, P.O. Box 8766, Saddar, Karachi - Pakistan.
Phones : 5688791, 5689285 Fax : 5689860
pjms@pjms.com.pk