|
|
||||
|
Published by : PROFESSIONAL MEDICAL PUBLICATIONS |
||||
|
ISSN 1681-715X |
||||
|
||||
|
- |
||||
|
ORIGINAL ARTICLE |
||||
|
- |
||||
|
Volume 23 |
October - December 2007 (Part-II) |
Number 6 |
||
|
|
||||
|
|
||||
|
|
||||
|
Published by : PROFESSIONAL MEDICAL PUBLICATIONS |
||||
|
ISSN 1681-715X |
||||
|
||||
|
- |
||||
|
ORIGINAL ARTICLE |
||||
|
- |
||||
|
Volume 23 |
October - December 2007 (Part-II) |
Number 6 |
||
|
|
||||
|
|
||||
Human Chorionic Gonadotropin (hCG):
A treatment of oligospermia
Mazhar Mushtaq1, Saghir Ahmed Jafri2,
Abdus Salam Sheikh3,
Sadia Ahmad4, Farah Deeba5, Khurram Salam6
Abstract
The most commonly accepted definition of the term infertility is the lack of pregnancy (regardless of cause) after one year of unprotected intercourse. Infertility affects approximately 15% of couples of reproductive age. Its prevalence has been stable during the past 50 years, although a shift in etiology and in the age of the patient has occurred. In Pakistan there are roughly 10-15% newly married couples who require some kind of medical help before they can become parents. Even if the wife is fertile and the husband is normal, there is roughly 1 in 3 chances the wife will conceive. A total of 17 patients were diagnosed for Oligospermia and were placed in one group. 5000 IU of hCG were administered intramuscularly once a week for four month. Every month serum testosterone and semen count was analyzed. Progressive increase of semen count was observed in four month. hCG though, never produced in male but has significant effect on spermatogenesis via LH receptor. Initial value of 25 million/ml roused to 69 million/ml in just four month. Use of hCG in male infertility is though common world wide but it has not gained its popularity in Pakistan. General awareness of its usage especially among physicians treating infertility is required.
KEY WORDS: Infertility, Fertility, Oligospermia, hCG, LH, FSH, Testosterone, Semen count.
Abbreviations: Human Chorionic Gonadotropin (hCG); Luteinizing Hormone (LH); Follical Stimulating Hormone (FSH).
Pak J Med Sci October - December 2007 (Part-II) Vol. 23 No. 6 840-846
1. Dr. Mazhar Mushtaq,
2. Prof. Saghir Ahmed Jafri,
Director,
Institute of Molecular Biology and Biotechnology,
3. Prof. Abdus Salam Sheikh,
Head of Biochemistry Department,
University College of Medicine,
4. Dr. Sadia Ahmad,
5. Dr. Farah Deeba,
6. Dr. Khurram Salam
1,4-6: Senior Lecturer,
Biochemistry Department,
University College of Medicine,
1-6: The University of Lahore
Lahore � Pakistan.
Correspondence
Dr. Mazhar Mushtaq,
E-mail: dr_hcg@yahoo.com
* Received for Publication: July 9, 2007
* Revision Received: October 2, 2007
* Revision Accepted: October 3, 2007
INTRODUCTION
The initial evaluation of the male patient should be rapid, noninvasive and cost-effective, as nearly 70% of conditions causing infertility in men can be diagnosed by personal history, physical examination, testicular volume estimation, hormonal and semen analysis. Further evaluation can then be initiated if necessary. A rational approach is necessary to perform the appropriate workup and to choose the best treatment options for the couple.
1Improvements in infertility treatment have made it possible for many patients to have a family. These new and advanced technologies include, In Vitro Fertilization (IVF), Intracytoplasmic Sperm Injection (ICSI), and other related procedures like Assisted Reproductive Technologies (ART). Other than these author wishes to introduce three new terms to this vocabulary, Artificially Induced Pregnancy (AIP); Bed Induced Pregnancy (BIP) and Chemical Induced Pregnancy (CIP).
Various treatments are available for the infertile couple, ranging from optimizing the current semen parameters with medical therapy to minor surgical procedures and finally to complex sperm retrieval like ART. Technological advancements in assisted reproduction make conceiving a child possible with viable sperm and egg. While the workup has traditionally been delayed until a couple has been unable to conceive for 12 months, beginning the workup at the first visit is now recommended.
2The pituitary gland, which lies on a stalk beneath the hypothalamus in the sella turcica, contains the gonadotropic cells that produce both FSH and LH. These are glycopeptides with a molecular weight of 10,000 daltons. They are made up of an alpha chain that is identical with that of hCG and Thyroid Stimulating Hormone (TSH) and a beta chain that is unique for each. FSH has a lower plasma concentration and longer half-life than LH, and it has less obvious pulsatile changes.
3The pulsatile nature of Gonadotropin Releasing Hormone (GnRH) is essential for normal gonadotropin release; a continuous stimulation inhibits their secretion.
4 After release into the systemic circulation, FSH and LH exert their effect by binding to plasma membrane receptors of the target cells. LH mainly functions to stimulate testosterone secretion from the Leydig cells of the testicle, while FSH stimulates Sertoli cells to facilitate germ cell differentiation.5 Gonadotropin release is modulated by a variety of other signals, such as estradiol (a potent inhibitor of both LH and FSH release), and inhibin from the Sertoli cell, which causes a selective decrease in FSH release.4 Decrease amount of LH production fail to initiate spermatogenesis thus causing oligospermia due to secondary deficiency of testosterone.6Use of hCG in such patient have become routine however in male endogenous production of hCG does not occur and is thus substituted from outside. Purified hCG is an effective substitute for LH since the structural homology between these two hormones which act through the same Leydig cell receptor.
7In any infertility work-up, both male and female partners are tested if pregnancy fails to occur after a year of regular unprotected sexual intercourse. It should be done earlier if a woman is over 35 years old or if either partner has known risk factors for infertility. A work-up can not only uncover the causes of infertility but detect other potentially serious medical problems as well, including genetic mutations, cancer or diabetes.
8Objectives: Main purpose of this study was to identify subjects with infertility due to; Normal or decreased LH level but decreased quantity of sperm count that is oligospermia. After complete scrutinizing these subjects would be given hormones viz, hCG, to see the effect on spermatogenesis. Identification of these subject and the protocols for the administration of hormone are described in the methodology.
SUBJECTS AND METHODS
The subjects were selected by simple random sampling technique. Couples with complaint of male infertility were contacted. Detailed history was taken from every patient and only those were selected who were willing to give their written consent and would fulfill the inclusion criteria. Study was approved by the medical ethics committee of The University of Lahore. All the work was supervised by the Director of "Institute of Molecular Biology and Biotechnology" (IMBB).
Settings: This study was planned to study the:
1. Best mode of diagnosis of male infertility.
2. Possible treatment of choice for male infertility.
3. Role of hCG in treatment of male infertility.
Inclusion Criteria: Following protocols were observed as inclusion criteria;
1. Infertile patient having oligospermia (Sperm count < 30 Million/ml).
2. Infertile oligospermic patient with history of delayed puberty.
3. Healthy, men aged 17�45 years living in a stable relationship and desiring fertility.
4. Male partner was asked to submit the OK report of female partner prior to entry in study.
5. Patient willing to provide written consent.
Exclusion Criteria: Following protocols were observed for infertile male;
1. Patient having history of mumps, orchitis, trauma to testis, excessive radiation to testis and uncorrected bilat cryptorchidism.
2. Patient on cytotoxic drugs, alcohol and spirolactone.
3. Patient of alpha blockers and on Anti Tuberculosis Treatment.
4. Patient suffering from autoimmune diseases or paraplegia.
5. Previous treatment of infertility specially use of hCG.
6. Couples with repeated history of missed abortion.
7. Any pathology with female partner.
Grouping: Patient with decreased sperm count < 30 Million/ml presenting Oligospermia or Azospermia as cause of infertility were of age 22-45 of different socio-economic status; all were placed in one group irrespective of their age or other factors.
Sampling procedures: Following protocols were observed to collect the semen and blood sample;
Each subject was asked to abstain for 48 � 72 hour prior to their visit to the infertility center and was asked to collect the semen sample in glass container. The sample was then analyzed for physical examination and microscopic examination. Blood sample were taken only at the initial visit for the hormonal level of FSH, LH, Prolectin and Testosterone.
Blood Sample Analysis: Hormonal level in blood sample was analyzed by using standard commercials kits available in the market. Hormones that were taken in to account were FSH, LH, Testosterone and Prolactin.

Semen Sample Analysis: Physical examination and Microscopic examination of semen sample were performed in routine laboratory.
Method for Data Collection: All the data was put in the spread sheet of Microsoft Office Excel version 2003.
Analysis of the result: Final result was evaluated by measuring P values with Microsoft Office Excel version 2003. P value indicates the probability of difference between two mean being significantly different or not. When P is > 0.05 the values is regarded as non significant and if <0.05 result are significant.
All patients were asked to closely observe any unwanted/beneficial effects. An unwanted effect includes; headache, blurred vision.
9 Nipple tenderness (Gynacomastia), testicular tenderness and increase/decrease testicular size.10 Beneficial effects includes; Enhanced libido and sex performance11 and weight loss.12RESULTS
The major dependent variable in the study was sperm count and independent variable was serum testosterone. Testosterone being primarily spermatogenesis variable was required to measure its level accurately as far as possible. Testosterone level and Sperm Count at 0 day of the treatment clearly gives inside clue for the importance of testosterone for spermatogenesis. However the availability of the other hormone required for the maturation of sperm are also significantly important.

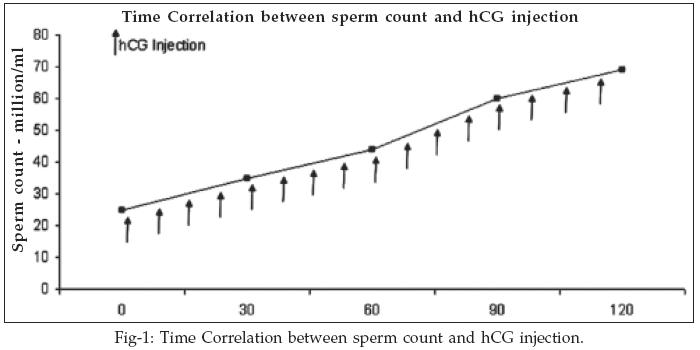
Dosage of hCG is marked with sign (). Which mean weakly injections of hCG is administered in divided dose of 5000IU each week intramuscularly for four month.
13 Sperm count was measured after each month showing progressive increase in their number each month. Seventeen patients were in this group each of them had Oligospermia as it was seen from their sperm count during the screening procedure. Progressive use of hCG in four month produces significant changes in the quantity of sperm count with initial average value of 25 million/ml rising to average 69 million/ml in four month. All of the 17 patients had normal levels of other hormone except one patient whose prolactin was elevated to 36ng/ml.

Use of hCG in these patient mimics the effect of LH by binding to LH receptor which in turn activate the cyclic AMP cascade and finally resulting in the synthesis of testosterone which is primary hormone for the spermatogenesis. Thus hCG results in the increased level of testosterone which in turn potentiate the spermatogenesis. Significant increase is seen with initial level of 4.17ng/ml and then rising to 10.35ng/ml in four month of hCG treatment. In the study four patients had initial semen count >30 million/ml, however they were unable to fertilize the ova of their partner for the past 13-15 month of their marriage.
Table-III shows the sperm count of every 30 days of treatment. After every 30 days P value does not show significant rise in sperm count however the over all increase of P value is moderately significant indicating successful induction of spermatogenesis by hCG. Once the induction start it continues to maintain the spermatogenesis thus producing significant result as we can see that in the P value which decreases as we move from Day 1 to Day 120 of treatment. Very shrewd question is frequently asked as this point of the study when spermatogenesis has been successfully induced. "Can the testosterone be substituted for hCG at this stage"? Replacing testosterone at any stage is NOT beneficial because oligospermia/azoospermia reappeared when hCG is substituted with testosterone.14

Mode of Action of hCG: The mechanism of hormone action is generally determined by its water solubility. Hydrophilic hormones tend to bind to receptors on the outer surface of cell membranes and activate 2nd messenger systems such as cAMP or alter membrane permeability.
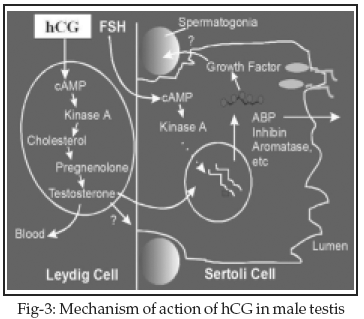
15 Those hormones that act through a 2nd messenger generally go on to influence the rate of enzymatic reactions within the cell. LH being water soluble acts through 2nd messenger system (Fig-2). In males, LH stimulates the interstitial cells of the testes to secrete testosterone which in return is the important component of spermatogenesis.15 Mechanism of action of hCG is the same as of LH (Fig-3). Structurally � unit of both these glycoprotein hormones exhibit 80% of homology hence binding of hCG and LH on the same receptor and there fore rendering the same mechanism of action. In short hCG mimics the activity of LH the only difference being the half life of hCG longer.15DISCUSSION
To procreate is natural instinct. If couple is not able to reproduce it gets socially and psychologically disturbed. Couples take it so stressfully that every sphere of couple�s life becomes affected. Diagnosis of such couple is far more important and remains an important issue in different communities. Similar social taboos are observed in Pakistan in some regions where male does not go for semen analysis in case of infertility but the male partner marries twice or thrice for being issueless.
hCG is normally produced in the female and thus having critical importance during pregnancy has been widely studied hormone at the molecular level. Patient having normal/low LH level and hence normal/low testosterone are suitable candidate for this study. hCG having advantage of longer time of action produces increased amount of testosterone than would LH, hence allowing its single dose in a week.
16 This hormone stimulates Leyding cell in the testis producing testosterone.Total 17 patients from different socioeconomic status were put in one group on the bases of decreased sperm count and relatively low testosterone level. These patients were given 5000 IU of hCG weekly for four month. Dependent and independent variables were checked after each month. P value showed significant differences. Though the sample size is not very large, still the results are convincing showing strong association of hCG in spermatogenesis. Use of hCG for the treatment of male infertility in human was reported in 1973 by Zarata.
17 He demonstrated the use of hCG in male just because male had poor semen quantity and quality with out the demonstration of the hormone level.The primary function of hCG is to increase the synthesis of testosterone as we see in (Fig-1). In our study exponential increase of 5.1ng/ml is observed which corresponds to the study of Bouloux where 4.52ng/ml is observed.
18 Testosterone thus produced results in the increase sperm concentration with mean increase of 45million/ml in 120 days as noted in our study. Thus the primary efficacy and end point was sperm density and semen volume after hCG treatment. These results coincides with Higashi who in 1998 studied 28 men with primary, complete isolated hypogonadotropic hypogonadism and administered hCG for 3-6 months followed by semen analysis and pregnancy in the female partner for couple who desired fertility.19Frequently hCG administration is accompanied by an increase in libido and improved sexual performance.
11 This also correlates with our study where 90 to 95% of patients had given positive response for their improved sexual activity. The primary end point was the increase sperm count and secondary end point is increased sexual performance were significant in our study. No serious study drug related adverse event like; blurred vision and scotomas11 was recorded except one patient who was dropped at early stage of study because he developed transient nipple tenderness and gynecomastia just after two injections which might have pre-existed and hCG have been predisposing factor.Treatment of infertility varies from region to region; botanical medicines are widely used in china and its peripheral region,
20 vitamins C21 and E22 can also solve the infertility problem. Problem remains with the diagnosis of infertility where hormone level plays variable role in its diagnosis. Disturbance of hormone level absolutely requires their substitution in combination or alone. Thus it is recommended that when labeling the male with infertility hormones level must be accessed beside sperm count. Though the usage hCG is not new but it has not gain its importance yet in Pakistan. 90% Physicians treating infertility are unaware of its usage.Recommendations
1. Public awareness for the male infertility, so that female is not the target every time when couple is issueless. Rural and tribal area is the most effected areas of such dilemma.
2. Physician awareness of the fact that which subjects are to label as infertility. As mentioned earlier semen count should NOT be the solo criteria to label the individual with infertility. Level of hormonal evaluation should never be compromised.
3. Strict and thorough history, physical examination and dwelling life style can be the key in the diagnosis of the cause of infertility.
4. Treating infertility not necessarily means pregnancy, however all the effort should be made for the conception. Follow up is an important issue where most of the patient does not comply.
5. Use of hCG is not new remedy for infertility but so far this hormone have failed to gain its popularity in south eastern Asia the reason being physicians treating infertility are unaware of its usage at least in Pakistan.
CONCLUSION
The objective of our study was to elucidate the usage of hCG in infertility mainly in oligospermic male; stress was also laid on their diagnosis which included hormone level and sperm count. Thus decreased sperm count only accounts for infertility but what type of infertility is diagnosed from hormonal level. Clinicians are thus advised to have all the parameter checked before labeling the patient with "infertility". Once the diagnosis is made the relevant treatment may then be commenced.
REFERENCES
1. Spitz A, Kim Ed, Lipshultz LI. Contemporary approach to the male infertility evaluation. Obst. Gynecol Clin North Am 2000;27(3):487-516.
2. Lunenfeld B, Olchovsky D, Tadir Y, Glezerman M. Treatment of male infertility with human gonadotrophins: selection of cases, management and results. Andrologia 1979;11(5):331-6.
3. Adamopoulos Da, Nicopoulou S, Kapoola N, Vassilopoulos P, Karamertzanis M. Testicula secretion after pulsatile human menopausal gonadotropin therapy in gonadotropin releasing hormone agonist desensitized dysspermic men. Fertil Steril 1994;62(1):155-61.
4. Glass AR, Vigersky RA. Leyding cell function in idiopathic oligospermia. Fertil Steril. 1980;34(2):144-8.
5. McLachlan RI, Finkel DM, Bremner WJ, Snyder PJ. Serum inhbin concentration before and during gonadotropin treatment in men with hypogonadotropic hypogonadism: Physiological and clinical implication. J Clin Endocrinol Metab 1990;70(5):1414-9.
6. Shane JM. Evaluation and treatment of infertility. Clinical Symptoms 1993;45:2.
7. Masala A, Delitala G, Alagna S, DeVilla L, Rovasio PP, Borroni G. Effect of synthetic LH-RH and hCG administration on plasma testosterone, rostenedione and esteradiol 17 beta levels in normal men and in patients with idiopathic oligospermia. Int J Fertil 1979;24(1):71-3.
8. Conway GS. Clinical manifestations of genetic defects affecting gonadotrophins and their receptors. Clin Endocrinol (Oxf) 1996;45:657-63.
9. Lee M, Fried WI, Sharifi R. Ocular adverse effects of human chorionic gonadotropin. Minerva Med 1983;74(19):1077-96.
10. Danielli L, Serour F, Brook I. The chorionic gonadotropin hormone: Possible cause of seminoma? J Gynecol Obstet Biol Reprod 1986;15(2):185-6.
11. Amelar RD, Dubin L, Croay JK. Human chorionic gonadotropin therapy in male infertility. JAMA 1977;237(22):2423.
12. Gusman H. Human Chorionic Gonadotropin in Obesity. Am J Clin Nutrition. 1969;22(6):686-95.
13. Fahmy I, Kamal A, Shamolul R, Mansour R, Aboulghar M. ICSI using testicular sperm in male hypogonadotropic hypogonadism unresponsive to gonadotrophin therapy. J Urol 2005;173(2):546.
14. Martin DU, Aubert ML, Biondo M, Campana A. Effect of gonadotrophins, testosterone and growth hormone on spermatogenesis in hypophysectomy patients. J Gynecol Obstet Bio Reprod (Paris) 1995;24(4):357-61.
15. Jennifer A. Mark S, Charles J, Allan M, David J. Luteinizing Hormone Receptor Mediated Effects on Initiation of Spermatogenesis in Gonadotropin deficient Mice. Biology of Reproduction 2004;70:32-8.
16. David K. Monash Institute of Reproduction and Development, Monash University, Hormonal Causes of Male Infertility. Andrology Australia 2001;1-2.
17. Zarate A, Valdes F, Gonzalez A, Perez Ubierna C, Canales ES, Schally A. Male Infertility. Fertil Steril 1973;24:485-6.
18. Bouloux PM, Burger HG, Skakkebaek NE, Baker GH, Conway AJ. Induction of spermatogenesis in hypogonadotropic azoospermic men who failed to respond to hCG. J Androl 2003;24(4):604-11.
19. Higashi Y, Takagi K, oshida K, Oshima H. Efficacy and safety of highly purified FSH with hCG for treating isolated hypogonadotropic hypogonadism. Fertil Steril 1998;70(2):256-62.
20. Chen JC, Xu MX, Chen LD. Effect of Panax notoginseng extracts on inferior sperm motility in vitro. Am J Chin Med 1999;27:123-8.
21. Dawson EB, Harris WA, Teter MC, Powell LC. Effect of ascorbic acid supplementation on the sperm quality of smokers. Fertil Steril 1992;58:1034-9.
22. Suleiman SA, Ali ME, Zaki ZM. Lipid peroxidation and human sperm motility: Protective role of vitamin E. J Androl 1996;17:530-7.
HOME | SEARCH | CURRENT ISSUE | PAST ISSUES
Professional
Medical Publications
Room No. 522, 5th Floor, Panorama Centre
Building No. 2, P.O. Box 8766, Saddar, Karachi - Pakistan.
Phones : 5688791, 5689285 Fax : 5689860
pjms@pjms.com.pk