|
|
||||
|
Published by : PROFESSIONAL MEDICAL PUBLICATIONS |
||||
|
ISSN 1681-715X |
||||
|
||||
|
- |
||||
|
ORIGINAL ARTICLE |
||||
|
- |
||||
|
Volume 24 |
April - June 2008 (Part-I) |
Number 2 |
||
|
|
||||
|
|
||||
|
|
||||
|
Published by : PROFESSIONAL MEDICAL PUBLICATIONS |
||||
|
ISSN 1681-715X |
||||
|
||||
|
- |
||||
|
ORIGINAL ARTICLE |
||||
|
- |
||||
|
Volume 24 |
April - June 2008 (Part-I) |
Number 2 |
||
|
|
||||
|
|
||||
Apolipoprotein E polymorphism, paraoxonase-1
activity and coronary artery disease: is there a link
F. Nabatchian1, Sh. Khaghani2, R. Miri3, M. Mahmoodi4, A. Karimi5, P. Pasalar6
ABSTRACT
Objective: The association of ApoE allele frequencies with coronary artery disease (CAD) remains unknown in developing countries. Paraoxonase 1 (PON1) is an enzyme that is associated with high density lipoprotein (HDL) and hydrolyzes oxidized lipids in oxidized low density lipoprotein (LDL) and thus protects against the development of atherosclerosis. In this study the association of apoE polymorphism and PON1 with premature CAD was determined.
Methodology: The frequency of apo E alleles and the activity of the PON1 among 162 patients with early and late onset CAD was examined.
Results: The mean age in the early and late-onset groups were 44.9 ± 4.9 and 71.7±2.5 years respectively. Patients with early-onset CAD had significantly lower serum levels of HDL-C and higher LDL-C/HDL-C ratio than the late-onset group. There was no difference in serum levels of triglyceride, total cholesterol and LDL-C between the two groups. The e2 allele was significantly higher in the late-onset group. 35.0% of early-onset and 17.5% of late-onset group had low level of PON1. 86% of the early-onset CAD and all of the late-onset CAD patients with e3/ e4 genotype, had low or moderate level of PON1 activity.
Conclusion: We found a positive association between PON1 activity, HDL level and the onset of CAD. Our study suggests that there are modest associations between CAD and apo E alleles and that the lower activity of PON1 in the presence of lower apo E level may increase the susceptibility to early-onset CAD.
KEY WORDS:
Paraoxonase, Atherosclerosis, Early-onset coronary artery disease, Apolipoprotein E polymorphism, Lipid peroxidation, Oxidative stress, Antioxidant.Pak J Med Sci April - June 2008 (Part-I) Vol. 24 No. 2 204-208
1. F. Nabatchian,
2. Sh. Khaghani,
3. P. Pasalar,
Department of Biochemistry,
4. R. Miri,
Hospital of Imam Hossein,
Shahid Beheshti University of Medical Sciences,
Tehran, Iran,
5. M. Mahmoodi,
Amiraalam Hospital,
1-3,5: Tehran University of Medical Sciences,
Tehran, Iran.
6. Karimi A,
Tehran Heart Center,
Tehran, Iran.
Correspondence
Parvin Pasalar, Ph.D
Clinical Biochemistry, Dept. of Biochemistry
Faculty of Medicine,
Tehran University of Medical Sciences,
Tehran, Iran
E-mail: pasalar@tums.ac.ir
* Received for Publication: December 9, 2007
* Revision Received: February 14, 2007
* 2nd Revision Received: September 29, 2007
* Final Revision Accepted: December 25, 2007
INTRODUCTION
Coronary artery disease (CAD) is a multifactorial disorder that has both genetic and nongenetic influences. The genetic influence is particularly strong in early onset CAD as shown in family and twin studies. The familial component appears to decline with advancing age and becomes negligible in patients presenting with CHD later in life.
1 In many studies, the genetic etiology of early-onset CAD has been attributed to genetic factors such as apolipoprotein E (apoE) genotypes.2Apo E gene is polymorphic with three common alleles,
e2, e3, and e4, producing three isoforms of the protein, E2, E3, and E4.3,4 e2 is associated with higher and e4 with lower concentrations of apo E.5 This polymorphism is consistently associated with plasma concentrations of total and LDL cholesterol.6 It has been shown that apo E knockout mice acquire increased susceptibility of lipid peroxidation under oxidative stress.7,8 The apo E genotype yields poor predictive values when screening for clinically defined atherosclerosis.2The other protein that has been associated with CAD is Paraoxonase (PON1). PON1 is exclusively associated with HDL and protects LDL from oxidation and prevents atherosclerosis development
9-11 via reduction of LDL in expense of PON1 oxidation (inactivation).12,13 The idea that PON1 should have antioxidant potential arose from its structure; rich in reducing amino acid cysteine.14,15 Increased susceptibility to atherosclerosis observed in PON1 knockout mice.16 Clinically, PON1 activity is low when pathological vascular ageing occurs early and when cardiovascular risk is high, however, its activity is highly variable and its regulation is complex, involving both genetic and environmental factors.17The interactive effect of PON1 and apo E was found in the development of the two most common forms of dementias; Alzheimer’s and vascular dementias.
18,19 Also it has been shown that the modification of the apo E binding site to the LDL receptor and proteoglycan might have caused an accumulation of oxidized VLDL in the circulation system.8Given the association between apo E gene polymorphism and apo E protein level, accelerated atherosclerosis in the absence of apo E, increased PON1 activity and protection of LDL from
oxidation and atherosclerosis development,15 we hypothesized that not only apo E polymorphism may be a risk factor for CAD but also its interaction of it with PON1 activity may be important in oxidative damages of endothelium and hence in atherogenesis. Accordingly, we hypothesized that because the genetic influence is stronger in early onset disease, patients presenting with early onset CAD would have an increased oxidative stress, compared to those manifesting CAD later in life. The delayed-onset was considered a sign of longevity and would serve as a comparative group to assess prevalence of the biochemical and genetic risk factors. In this study we measured total cholesterol (TC), HDL (HDL-C), LDL cholesterol (LDL-C), triglyceride (TG) and PON1 activity in early and late-onset CAD patients. We also studied and compared the frequency of the apo E alleles and explored synergies between different apo E alleles and PON1 activity in the two groups.METHODOLOGY
2.1 Study subjects: The patients were recruited from the cardiac wards of Tehran University hospitals. All had CAD confirmed by angiography demonstrating one or more epicardial coronary vessels with greater than 50% diameter stenosis and were on optimal medical therapy. Patients with diabetes mellitus, kidney, thyroid or liver disease were excluded. Eligible patients were sequentially recruited into two groups: early-onset CAD (n = 80) were those presenting with CAD under the age of 55 years (mean Age = 44.9± 4.9 years); late-onset CAD (n = 80) were patients presenting with CAD for the first time over the age of 65 years (mean Age = 71.7 ± 2.5 years). The study was conducted following the approval of our institutional review board, and informed consent was obtained from all patients before inclusion in the study.
2.2. Laboratory methods
2.2.1. Lipid profile and PON1 analysis: A blood specimen was collected after overnight fasting for 12 hour. Serum TC and TG levels were measured by commercially available enzymatic kits. HDL-C was measured after precipitation with phosphotungstic acid and LDL-C was calculated using Friedwald’s
20 formula for serum samples with TG values less than 400mg/dl. In patients with a TG value greater than 400mg/dl, LDL-C levels were not calculated. PON1 activity was measured using paraoxon as the substrate.21 Paraoxanase activity is defined as the amount of enzyme which converts 1m mol paraoxone (Aryldialkyl phosphate) to nitrophenol in one minute.2.2.2. Genotyping: Genomic DNA was extracted from whole blood leukocytes by a standard method.
22 Apo E genotyping was performed using 5'-TAAGCTTGGCACGGCTGT CCAAGGA-3' (forward) and 5'-ACAGAAT TCGCCCCGGCCTGGTACAC-3' (reverse) primers.23 In this methd PCR product was incubated overnight at 37°C with cfoI restriction endonuclease. The digestion products were then separated using PAGE (15%) for 1.5 hours at 180 volts. The gel was then silver stained.3. Statistical analysis: Statistically significant differences between parameters with a gaussian distribution (age, serum-TC, LDL-C and HDL-C) were sought by Student’s unpaired t-test. Variables with a non-gaussian distribution (serum TG and PON1 activity) were compared using the Mann-Whitney U test. The ÷2 test was used to determine the significance of differences in allele frequency. P<0.05 were considered significant.
RESULTS
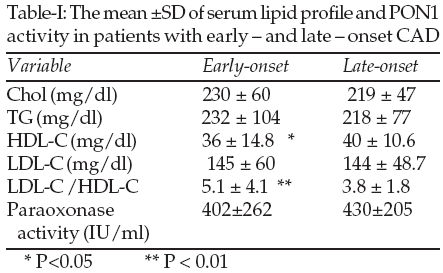
Patients with early-onset CAD had significantly lower serum levels of HDL-C (P<0.05) and higher LDL-C/HDL-C ratio (P<0.01) than the late-onset group. There was no difference in serum levels of TG, TC, and LDL-C between the two groups. The lipid profile of the two groups is shown in Table-I.
The frequency of the e
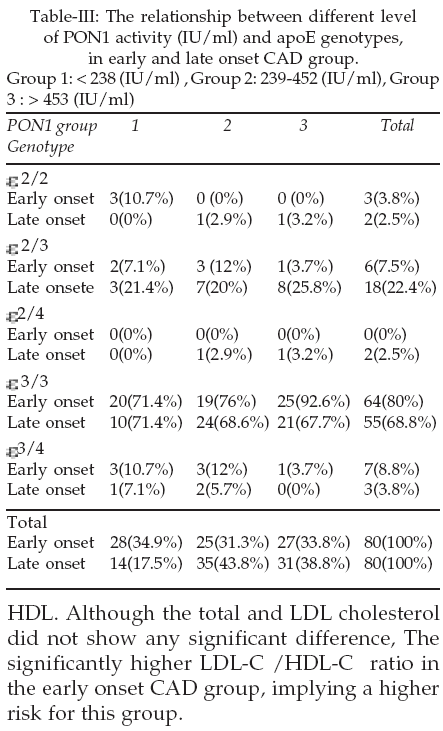
3 allele was 88.1 and 81.9 percent in early and late onset CAD group respectively with no significant difference (Table-II). The frequency of e2 allele was 7.5 and 15 percent in early and late onset CAD group respectively with significant difference.

The prevalence of e2/ e3 genotype was 7.5 and 22.4% in early and late –onset CAD respectively, with a significant difference (Table-III). The prevalence of e3/ e4 genotype was significantly different in the two groups. Although PON1 activity was not significantly different, when we divided the patients of each group into three groups of low, moderate and high level of PON1 activity, we found that 35.0% of early-onset group had the low level of PON 1 whereas this percentage was 17.5% in late-onset group. 86% of the early-onset CAD and all of the patients of late-onset CAD with apoe3/ e4 genotype, had low or moderate level of PON1 activity.
DISCUSSION
Apo E influences lipoprotein metabolism and confers a risk for CHD. In accordance with other studies
24,25 we found that e3 is the most frequent and e4 is the least frequent allele in Iranian population (Table-II). The significantly increased prevalence of e2 allele in our older group with CAD (15 vs.7.5%) is in accordance with other studies demonstrating that the e2 is found more frequently in older compared to younger control groups.26 The reason for the higher prevalence of e2 allele in the older population is not clear, but since apoe2 is associated with higher concentrations of apo E5,6 and the apo E knockout mice show accelerated atherosclerosis lipid peroxidation7 it could be speculated that higher concentrations of apo E5,6 may delay the onset of CAD until later in life.
The low level of PON1 activity in the early-onset CAD group was two fold greater than the late-onset, which is consistent with studies, including Tward et al27 who demonstrated that PON1 transgenic mice are at lower risk for atherosclerosis and Rozenberg16 who showed PON1 knockout mice are more prone to the disease. Because HDL3 has the highest cardio protective effects and PON1 activity28,13 we may speculate that cardio-vasoprotective effect of the HDL is due to its association with PON1. The significant lower level of PON1 activity and HDL-C in the early onset CAD group may somehow related to lower level of HDL. Although the total and LDL cholesterol did not show any significant difference, The significantly higher LDL-C /HDL-C ratio in the early onset CAD group, implying a higher risk for this group.
In our study, 86% of the early-onset CAD and all of the patients of late-onset CAD with e3/ e4 genotype, had low or moderate level of PON1 activity, since apoe4 is associated with lower concentrations of apo E5,6 this may imply that their combined antioxidant effect may somehow be cumulative. The interrelationship between PON1 activity and apo E has been shown by Shih et al29 who showed, increased atherosclerosis in mice, when PON1 deficiency was combined with apolipoprotein E (apoE) deficiency. Getz and Godfrey30 found that exogenous PON1 is able to reverse the oxidative stress in macrophages in aged apolipoprotein E deficient and PON1 deficient mice.
CONCLUSION
In summary, our study suggests that there are modest associations between CAD, apoE polymorphism and PON1 activity and that the lower activity of PON1 in the presence of lower level of apo E concentration may increase the susceptibility to atherosclerosis.
ACKNOWLEDGEMENTS
The authors thank the nurses and night shifts staffs from Amiraalam Hospital and Tehran Heart Center for assistance in preparation of the blood samples. The research council of Tehran University of Medical sciences grant number 1025 has supported this research.
REFERENCES
1. Rissanen A. "Familial occurrence of coronary heart disease: Effect of age at diagnosis Am J Cardiol 1979;44:60-6.
2. Wilson PW, Myers RH, Larson MG, Ordovas JM, Wolf PA, Shaefer EJ. "Apolipoprotein E alleles, dyslipidemia and coronary heart disease. The Framingham Offspring Study" JAMA 1994;272:1666-71.
3. Luo C C, Li WH, Moore MN. "Structure and evolution of the apolipoprotein multigene family" J Mol Biol 1986;187:325-40.
4. Weisgraber KH, Rall SC, Mahley RW. "Human E apoprotein heterogeneity: cysteine-arginine interchanges in the amino acid sequence of the apo E isoforms" J Biol Chem 1981;256:9077-83.
5. Hanis CL, Hewett-Emmett D, Douglas TC. "Effects of the apolipoprotein E polymorphism on levels of lipids, lipoproteins, and apolipoproteins among Mexican-Americans in Starr County, Texas" Arterioscler Thromb Vasc Biol 1991;11:362-70.
6. Siest G, Pillot T, Régis-Bailly A. "Apolipoprotein E: an important gene and protein to follow in laboratory medicine" Clin Chem 1995;41:1068-86.
7. Hayek T, Oiknine J, Brook JG, Aviram M. "Increased plasma and lipoprotein lipid peroxidation in apo E-deficient mice" Biochem Biophys Res Commun 1994;30:201(3):1567-74.
8. Arai H, Uchida K, Fukunaga K, Nagasaka Y, Mohri S, Furumoto H, et al. "Effect of alpha-tocopherol on the oxidative modification of apolipoprotein E in human very-low-density lipoprotein" Biosci Biotechnol Biochem 2003;67(2):402-5.
9. Hegle RA. "Paraoxonase genes and disease" Ann Med 1999;31(3):217-24.
10. Rosenblat M, Draganov D, Watson CE. "Mouse macrophage PON2 is increased whereas cellular PON3 activity is decreased under oxidative stress" Arterioscler. Thromb Vasc Biol 2003;23(3);468-74.
11. Noto H, Hashimoto Y, Satoh H. "Exclusive association of paraoxonase 1 with igh-density lipoprotein particles in apolipoprotein A-I deficiency" Biochem Biophys Res Commun 2001;289:395-401.
12. Aviram M, Rosenblat M, Billecke S. "Human serum paraoxonase (PON1) is inactivated by oxidized low density lipoprotein and preserved by antioxidants". Free Radic Biol Med 1999;26:892-904.
13. Zhang B, Fan P, Shimoji E. "Inhibition of Cholesteryl Ester Transfer Protein Activity by JTT-705 Increases Apolipoprotein E–Containing High-Density Lipoprotein and Favorably Affects the Function and Enzyme Composition of High-Density Lipoprotein in Rabbits" Arterioscler, Throm Vasc Biol 2004;24:1910-15.
14. Aviram M, Billecke S, Sorenson R. "Paraoxonase active site required for protection against LDL oxidation involves its free sulfhydryl group and is different from that required for its arylesterase/paraoxonase activities" Arterioscler Throm Vasc Biol 1998;18:e617-e1624.
15. Shih D M, Gu L, Xia Y R, "Mice lacking serum paraoxonase are susceptible to organophosphate toxicity and atherosclerosis", Nature 1998;394:284-7.
16. Rozenberg O, Shih DM, Aviram M. "Paraoxonase 1 (PON1) attenuates macrophage oxidative status: studies in PON1 transfected cells and in PON1 transgenic mice." Atherosclerosis 2005;181(1):9-18.
17. Bin Ali A, Zhang Q, Lim YK, Fang D, Retnam L, Lim SK. "Expression of major HDL-associated antioxidant PON-1 is gender dependent and regulated during inflammation" Free Radic Biol Med. 2003;1:34(7):824-9.
18. Gupta H, White CR, Handattu S, Garber DW, Datta G, Chaddha M, et al. "Apolipoprotein E mimetic Peptide dramatically lowers plasma cholesterol and restores endothelial function in watanabe heritable hyperlipidemic rabbits" Circulation, 2005;14;111(23):3112-8.
19. Janka Z, Juhasz A, Rimanoczy AA, Boda K, Marki-Zay J, et al. "Codon 311 (Cys —> Ser) polymorphism of paraoxonase-2 gene is associated with apolipoprotein E4 allele in both Alzheimer’s and vascular dementias" Mol Psychiatry, 2002;7(1):110-2.
20. Friedewald WT, Levy RI, Fredrickson DS. "Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge" Clin Chem 1972;18:499-502.
21. Mackness B, Mackness MI, Arrol S, Turkie W, Julier K, Abuasha B, et al. "Serum paraoxonase (PON1) 55 and 192 polymorphism and paraoxonase activity and concentration in non-insulin dependent diabetes mellitus" Atherosclerosis 1998;139(2):341-9.
22. Sambrook J, Russell DW. "Preparation and analysis of Eukaryotic genomic DNA" In: Molecular cloning: A laboratory manual edited by: Argentine J., Irwin N. Vol.1 Cold spring Harbor Laboratory Press; 2001; protocol 6.1.
23. Lahoz C, OsGood D. "Frequency of phenotype –genotype discrepancies at the apolipoprotein E locus in a large population study" Clin Chem 1996;42:11,1817-23.
24. Azizi F, Rahmani M, Raiszadeh F, Solati M, Navab M. "Association of lipids, lipoproteins, apolipoproteins and paraoxonase enzyme activity with premature coronary artery disease." Coron Artery Dis 2002;13(1):9-16.
25. Vaisi Raygani A, Zahrai M, Vaisi Raygani A, Doosti M, Javadi Ec, Rezaei M, et al. "Association between apolipoprotein E polymorphism and Alzheimer disease in Tehran, Iran" Neuro science Letters 2005;375(1)1-6.
26. Schachter F, Faure-Delanef L, Guenot F. "Genetic associations with human longevity at the APOE and ACE loci." Nature Genet 1994;6:29-32.
27. Tward A, Xia YR, Wang XP. "Decreased atherosclerotic lesion formation in human serum paraoxonase transgenic mice "Circulation, 2002;106:484-90.
28. Bergmeier C, Siekmeier R, Gross W. "Distribution spectrum of paraoxonase activity in HDL fractions." Clin Chem 2004;50(12):2309-15.
29. Shih DM, Xia YR, Wang YP. "Combined serum paraxonase knockout/apolipoprotein E knockout mice exhibit increased lipoprotein oxidation and atherosclerosis" J Biol Chem 2000;275:17527-35.
30. Getz GS, Reardon CA. "Paraoxonase, a cardio protective enzyme:ccontinuing issues. Curr Opin Lipidol 2004;15(3):261-7.
HOME | SEARCH | CURRENT ISSUE | PAST ISSUES
Professional
Medical Publications
Room No. 522, 5th Floor, Panorama Centre
Building No. 2, P.O. Box 8766, Saddar, Karachi - Pakistan.
Phones : 5688791, 5689285 Fax : 5689860
pjms@pjms.com.pk